Read A Brief Summary Of This Topic
William Thomson, Baron Kelvin, in full William Thomson, Baron Kelvin of Largs, also called Sir William Thomson, , Scottish engineer, mathematician, and physicist who profoundly influenced the scientific thought of his generation.
Thomson, who was knighted and raised to the peerage in recognition of his work in engineering and physics, was foremost among the small group of British scientists who helped lay the foundations of modern physics. His contributions to science included a major role in the development of the second law of thermodynamics the absolute temperature scale the dynamical theory of heat the mathematical analysis of electricity and magnetism, including the basic ideas for the electromagnetic theory of light the geophysical determination of the age of the Earth and fundamental work in hydrodynamics. His theoretical work on submarine telegraphy and his inventions for use on submarine cables aided Britain in capturing a preeminent place in world communication during the 19th century.
How Is Energy Measured In Physics
The standard unit used to measure energy and work done in physics is the joule which has the symbol J. In mechanics 1 joule is the energy transferred when a force of 1 Newton is applied to an object and moves it through a distance of 1 meter.The standard unit used to measure energy and work
work is the energy transferred to or from an object via the application of force along a displacement
Bringing These Ideas Into Your Classroom
As is illustrated in the TED-ed video, basic mathematical concepts can be used to understand how energy is quantified, such as measuring energy from two different forms, then converting those quantities into common units. Terms such as power , work can be easily measured and calculated. All of these terms have alternate, but related, meanings in daily life, so getting students familiar with the mathematical definitions will require students to understand slightly different meanings for the same words.
Many forms of energy transformation are directly observable in the classroom, so demonstrations are an effective means to illustrate transformations between different forms of energy.
Don’t Miss: Types Of Proofs In Geometry
Is Power A Scalar Quantity
Power is a scalar quantity because it is a ratio of two scalar quantities.
Watch this fun and engaging rapid fire session based on the topic Work, Energy and Power!
Work, Power and Energy is one of the important topics of JEE Main and JEE Advanced, watch the video and understand the type of questions asked from this topic!
Stay tuned to BYJUS and Fall in Love with Learning!
Put your understanding of this concept to test by answering a few MCQs. Click Start Quiz to begin!
Select the correct answer and click on the Finish buttonCheck your score and answers at the end of the quiz
Law Of Conservation Of Energy
The law of conservation of energy is one of the basic laws in physics. It governs the microscopic motion of individual atoms in a chemical reaction. The law of conservation of energy states that In a closed system, i.e., a system that is isolated from its surroundings, the total energy of the system is conserved. According to the law, the total energy in a system is conserved even though the transformation of energy occurs. Energy can neither be created nor destroyed, it can only be converted from one form to another.
Although there are many forms of energy, it is broadly categorized into:
Recommended Reading: Who Wrote The Early Textbook Principles Of Psychology
Students Can Experience And Recognize Different Forms Of Energy
Companion video by the Department of EnergyView a non-YouTube version of this video
Understanding how different types of energy are defined and measured offers a baseline from which to teach about other aspects of energy. The concepts of energy loss, energy transfer from one system to another, and ways to measure energy are essential concepts for teaching about energy. While it may be tempting to skip over these fundamentals and begin teaching about wind turbines and solar panels, it’s important to establish a frame of reference for understanding what energy is before discussing different fuels, sources of energy, and uses of energy. What is fascinating about energy is how one form of energy can be transformed into seemingly unrelated forms of energy. James Prescott Joule did pioneering experiments showing that a quantity of mechanical can be transformed into the same amount of thermal energy. For example, an explosion converts chemical potential energy into kinetic energy, radiant energy, and thermal energy. Radiant energy can be transformed into electrical energy by a photovoltaic cell. Thermal energy can be transformed into electrical energy by a thermo-electric generator.In all cases of energy transformation, some energy is transformed into thermal energy. Because this energy can often not be recovered in a useful way, this thermal energy is often considered to be wasted, or lost.
What Is The Relation Between Commercial And Si Unit Of Energy
The commercial unit of energy is 1 kWh. One kilowatt-hour is defined as the amount of energy consumed by a device in one working hour at a constant rate of one kilowatt.The SI unit of energy is joule.Therefore, the relationship between commercial and SI units of energy is:1 kWh = 1kW x 1h = 1000W x 1h = 1000 x 3600 s = 3.6 x106 J
Also Check: How Hard Is Ap Physics
Example Of Energies In Electronvolts
- Thermal neutrons are neutrons in thermal equilibrium with a surrounding medium of temperature 290K . Most probable energy at 17°C for Maxwellian distribution is 0.025 eV .
- Thermal energy of a molecule is at room temperature about 0.04 eV.
- Approximately 1 eV corresponds to an infrared of wavelength 1240 nm.
- Visible light photons have energies in range 1.65 eV 3.26 eV .
- The first resonance in n + 238U reaction is at 6.67 eV , which corresponds to the first virtual level in 239U, has a total width of only 0.027 eV, and the mean life of this state is 2.4×10-14s.
- Ionization energy of atomic hydrogen is 13.6 eV.
- Carbon-14 decays into nitrogen-14 through beta decay . The emitted beta particles have a maximum energy of 156 keV, while their weighted mean energy is 49 keV.
- High energy diagnostic medical x-ray photons have kinetic energies of about 200 keV.
- Thallium 208, which is one of nuclides in the 232U decay chain, emits gamma rays of 2.6 MeV which are very energetic and highly penetrating.
- Typical kinetic energy of alpha particle from radioactive decay is about 5 MeV. It is caused by the mechanism of their production.
- The total energy released in a reactor is about 210 MeV per 235U fission, distributed as shown in the table. In a reactor, the average recoverable energy per fission is about 200 MeV, being the total energy minus the energy of the energy of antineutrinos that are radiated away.
- Cosmic ray can have energies of 1 MeV 1000 TeV.
Cite This Page As Follows:
“What is the measure of an average kinetic energy?” eNotes Editorial, 28 Nov. 2008, https://www.enotes.com/homework-help/what-measure-an-average-kinetic-energy-44249.Accessed 12 Sep. 2022.
Who are the experts?Our certified Educators are real professors, teachers, and scholars who use their academic expertise to tackle your toughest questions. Educators go through a rigorous application process, and every answer they submit is reviewed by our in-house editorial team.
Kinetic Energy is the energy of motion. Another kind of energy, Potential, or Static Energy, is the energy of position. Both energies are understood to refer to force acting upon matter. Together, these energies add up to a system’s total energy, or
E = E + E
Potential and Kinetic are convertible, one to the other. If you consider the system of a book on a library floor, it has no Potential and no Kinetic However, picking it up and placing it on the library counter imparts…
You May Like: Is Anthrax Biological Or Chemical
Kinetic And Potential Energy
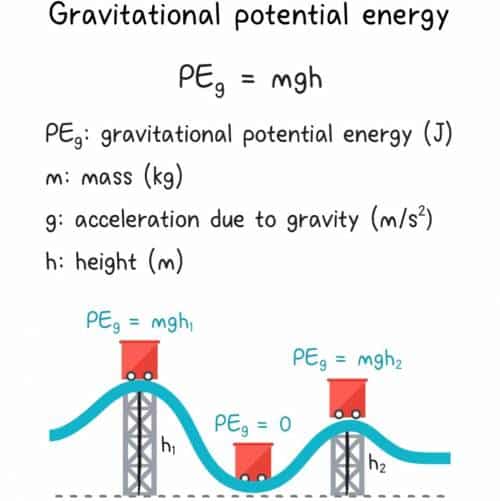
An object in motion possesses its energy of movement, which is equivalent to the work that would be required to bring it to rest. This is called its kinetic energy, and it is dependent on the square of the object’s velocity as well as one half of its mass . An object at rest in Earth’s gravitational field possesses potential energy by virtue of its altitude if it were to fall freely, it would gain kinetic energy equal to this potential energy. Potential energy is dependent on the object’s mass, its height and the acceleration due to gravity . Mathematically, this is:
Can Energy Be Created
Energy can be neither created nor destroyed. It can only be changed from one form to another. This principle is known as the conservation of energy.
energy, in physics, the capacity for doing work. It may exist in potential, kinetic, thermal, electrical, chemical, nuclear, or other various forms. There are, moreover, heat and worki.e., energy in the process of transfer from one body to another. After it has been transferred, energy is always designated according to its nature. Hence, heat transferred may become thermal energy, while work done may manifest itself in the form of mechanical energy.
All forms of energy are associated with motion. For example, any given body has kinetic energy if it is in motion. A tensioned device such as a bow or spring, though at rest, has the potential for creating motion it contains potential energy because of its configuration. Similarly, nuclear energy is potential energy because it results from the configuration of subatomic particles in the nucleus of an atom.
Energy can be converted from one form to another in various other ways. Usable mechanical or electrical energy is, for instance, produced by many kinds of devices, including fuel-burning heat engines, generators, batteries, fuel cells, and magnetohydrodynamic systems.
In the International System of Units , energy is measured in joules. One joule is equal to the work done by a one-newtonforce acting over a one-metre distance.
Also Check: What Are Human And Physical Features In Geography
British Imperial / Us Customary Units
The British imperial units and U.S. customary units for both energy and work include the foot-pound force , the British thermal unit which has various values in the region of 1055 J, the horsepower-hour , and the gasoline gallon equivalent .
| Learn how and when to remove this template message) |
A unit of electrical energy, particularly for utility bills, is the kilowatt-hour one kilowatt-hour is equivalent to 3.6 megajoule. Electricity usage is often given in units of kilowatt-hours per year or other time period. This is actually a measurement of average power consumption, meaning the average rate at which energy is transferred. One kilowatt-hour per year is about 0.11 watts.
Multiplication Of The Units Of Power With Units Of Time
When the Watt is multiplied by a unit of time, an energy unit is formed as follows: 1 Ws = 1 J. The use of the kilowatt-hour is more common: 1 kWh = 3600 kWs = 3.6 MJ. Besides the second and the hour , the day and the year are also used, with 1 yr = 365.2425 d = 31,556,952 s. So, for example, energy of one Megawatt-year can be written as 1 MWyr = 31.557952 TJ .The annual consumption of 1 toe/yr corresponds to the daily consumption of about 31.56 kWh/d.The annual consumption of 1 GJ/yr corresponds to the daily consumption of about 0.7605 kWh/d.
Don’t Miss: Which Statement About Social Psychology Is Most Accurate
Measuring Energy Vs Power
Although it is not possible to directly measure energy, the work done can be defined and measured. The methods involves using a calorimeter, which measures the heat absorbed or released in chemical reactions or physical changes, thermometer, which measures temperature or bolometer that is employed to measure the intensity of radiation. Energy generated can be stored whereas power cannot.
Since power is energy per unit of time, in theory it can be calculated after measuring the energy used per second. When calculating the real power consumption of an electrical device, it is essential to measure the voltage applied and the current consumed, taking into account the power that is dissipated in the circuit.
Atom Physics And Chemistry
In physics and chemistry, it is common to measure energy on the atomic scale in the non-SI, but convenient, units electronvolts . 1 eV is equivalent to the kinetic energy acquired by an electron in passing through a potential difference of 1 volt in a vacuum. It is common to use the SI magnitude prefixes with electronvolts. Because of the relativistic equivalence between mass and energy, the eV is also sometimes used as a unit of mass. The Hartree is commonly used in calculations. Historically Rydberg units have been used.
Recommended Reading: What Is Italy’s Geography
The Formula For Energy:
Kinetic Energy: The energy exists due to the motion of an object is known as Kinetic Energy. For example, a moving van, flowing water, etc.
Where,
| h | Height |
Mechanical Energy: It is the sum total of potential energy and kinetic energy which is the energy associated with the motion & position of any object. Therefore, the formula of mechanical energy will be:
Mechanical Energy = Kinetic Energy + Potential Energy
The law of conservation of energy is one of the basic laws of Physics. It states that In a closed and isolated system from its surroundings, the total energy of the system will be conserved. Thus energy can neither be created nor destroyed, although it can only be transformed from one form to another.
Energy And Classification Of Energy
In recent days, we often hear about energy. Every invention and civilization is based upon acquiring and effectively using energy. This is possible by the unique property of our Universe, that it can transfer and transform energy, but the total amount is always the same .
One fundamental focus of physics is to investigate energy. Energy in Physics can be generally defined as a scalar quantity associated with the state or condition of one or more objects.
Read Also: Transition To Algebra Unit 1 Answers
How Is Energy Measured
Understanding BTUs, therms, quads, and more.
A British Thermal Unit is a measure of energy content, usually used to describe the energy content of fuels. Because a Btu is so small, energy is usually measured in millions of Btus.
- 1 Btu = the amount of energy required to increase the temperature of one pound of water by one degree Fahrenheit. This is roughly the heat produced from burning one match.
- 1,000 Btu = four-fifths of the energy contained in a peanut butter and jelly sandwich.
- 100,000 Btu = one therm, such as what you would find on your gas bill. One therm is roughly equal to the energy contained in 80 peanut butter and jelly sandwiches. The average U.S. household used about 420 therms, or 42 million Btu, of natural gas in 2011 .
- 312 million Btu = U.S. per capita energy consumption in 2011 .
- 1 quadrillion Btu = one quad. The U.S. consumes more than 97 quads annually .
References
How To Calculate Power Using The Work And Power Calculator
For some real-life applications of power, check out the cycling wattage calculator!
Recommended Reading: Year 10 Coordinate Geometry Test
Energy Is A Word With Many Meanings Yet No Universal Definition
Provenance:Reuse:
Mechanical energy is is the energy of mechanical systems, such as a ball rolling on a ramp, or a marble fired from a slingshot. Mechanical energy can be in three forms:
- Gravitational potential energy is the energy of an object or system due to gravitational attraction. For example, we can calculate the mechanical energy of a ball that is going to be released from a high window, or the gravitational potential energy of the water in a reservoir used for hydropower.
- Kinetic energy is energy due to the motion of an object. A speeding car, a baseball lofting through the air, and a skier sliding downhill are all examples of objects with kinetic energy. Flywheels are a method of storing kinetic energy.
- Elastic potential energy is the energy stored in a stretched spring, rubber band, or other elastic material.
Thermal energy is the energy that results from kinetic energy of molecules of a substance. A hot tea kettle has more thermal energy than a cold one. Objects that feel warm are emitting thermal energy, and the transfer of thermal energy causes changes in temperature.
Radiant energy is the energy from electro-magnetic radiation, such as visible light, microwaves, or x-rays.
Chemical energy is energy stored in chemical bonds. Gasoline and food are examples of compounds with chemical potential energy.