How Are Photosynthesis And Cellular Respiration Connected
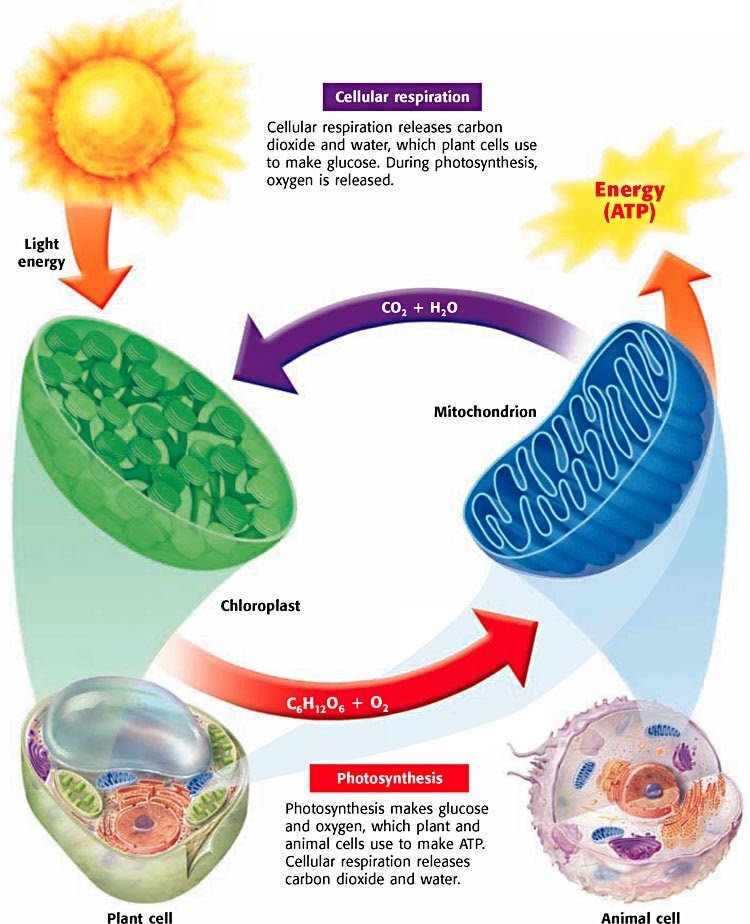
When I think about the connections between photosynthesis and cellular respiration, I cant help but start singing Circle of Life from The Lion King in my head. Why? Because the products of photosynthesis are required for cellular respiration, and the products of cellular respiration can be used to power photosynthesis.
Putting the chemical formulas for these processes side-by-side shows this quite clearly:
6CO2 + 6H2O + Sunlight C6H12O6 + 6O2
Cellular Respiration:C6H12O2 + 6O2 6CO2 + 6H2O + 38* ATP
*The number of ATP molecules produced can vary. 38 ATP is the theoretical maximum yield for the metabolism of one molecule of glucose.
The food that plants make and the waste product from producing that food give animals like us the materials we need to carry out aerobic cellular respiration. We breathe in the oxygen from the air and either eat plants or other animalseither way, plants and their delicious glucose are at the root of ourfood web. In return, humans and other organisms that carry out aerobic respiration put the waste products from this process back into the atmosphere.
Plants carry out both photosynthesis and cellular respiration. They make their own food, and then break down those glucose molecules later, generating ATP to power their cellular processes.
Before we go, heres a handy chart comparing photosynthesis and aerobic cellular respiration. Happy studying!
S In The Citric Acid Cycle
Step 1. Prior to the start of the first step, pyruvate oxidation must occur. Then, the first step of the cycle begins: This is a condensation step, combining the two-carbon acetyl group with a four-carbon oxaloacetate molecule to form a six-carbon molecule of citrate. CoA is bound to a sulfhydryl group and diffuses away to eventually combine with another acetyl group. This step is irreversible because it is highly exergonic. The rate of this reaction is controlled by negative feedback and the amount of ATP available. If ATP levels increase, the rate of this reaction decreases. If ATP is in short supply, the rate increases.
Step 2. In step two, citrate loses one water molecule and gains another as citrate is converted into its isomer, isocitrate.
Step 3. In step three, isocitrate is oxidized, producing a five-carbon molecule, -ketoglutarate, together with a molecule of CO2 and two electrons, which reduce NAD+ to NADH. This step is also regulated by negative feedback from ATP and NADH, and a positive effect of ADP.
Steps 3 and 4. Steps three and four are both oxidation and decarboxylation steps, which release electrons that reduce NAD+ to NADH and release carboxyl groups that form CO2 molecules. -Ketoglutarate is the product of step three, and a succinyl group is the product of step four. CoA binds the succinyl group to form succinyl CoA. The enzyme that catalyzes step four is regulated by feedback inhibition of ATP, succinyl CoA, and NADH.
First Half Of Glycolysis
Figure 2. The first half of glycolysis uses two ATP molecules in the phosphorylation of glucose, which is then split into two three-carbon molecules.
Step 1. The first step in glycolysis is catalyzed by hexokinase, an enzyme with broad specificity that catalyzes the phosphorylation of six-carbon sugars. Hexokinase phosphorylates glucose using ATP as the source of the phosphate, producing glucose-6-phosphate, a more reactive form of glucose. This reaction prevents the phosphorylated glucose molecule from continuing to interact with the GLUT proteins, and it can no longer leave the cell because the negatively charged phosphate will not allow it to cross the hydrophobic interior of the plasma membrane.
Step 2. In the second step of glycolysis, an isomerase converts glucose-6-phosphate into one of its isomers, fructose-6-phosphate. An isomerase is an enzyme that catalyzes the conversion of a molecule into one of its isomers. This change from phosphoglucose to phosphofructose allows the eventual split of the sugar into two three-carbon molecules.
Step 4. The newly added high-energy phosphates further destabilize fructose-1,6-bisphosphate. The fourth step in glycolysis employs an enzyme, aldolase, to cleave 1,6-bisphosphate into two three-carbon isomers: dihydroxyacetone-phosphate and glyceraldehyde-3-phosphate.
You May Like: Geometry Escape Challenge A Answer Key
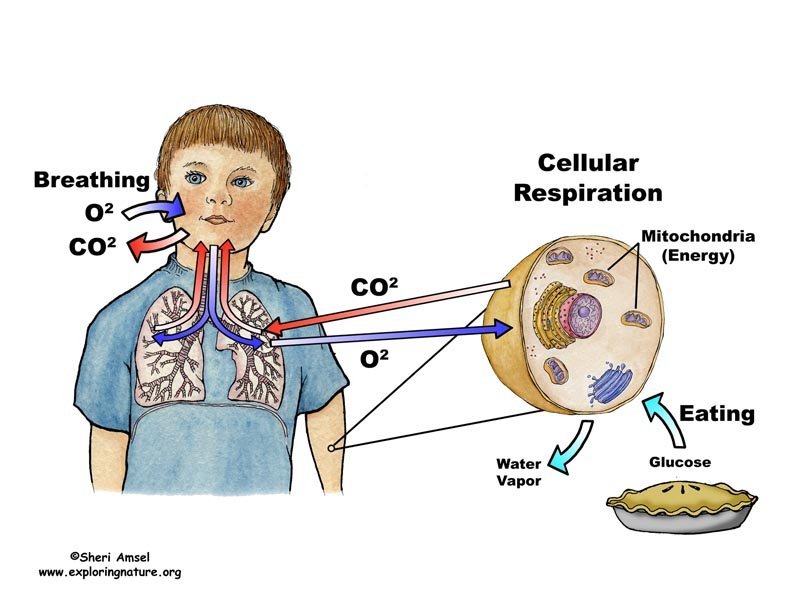
What Is Cellular Respiration
Cellular respiration is a set of metabolic reactions that take place in all living cells to release energy by converting biochemical energy from nutrients into adenosine triphosphate- ATP. Based on the oxygen demand, cellular respiration is divided into- Aerobic respiration and Anaerobic respiration.
Example Question #: Cellular Respiration
In comparison to fermentation, the aerobic pathways of glucose metabolism yield more __________.
Fermentation is the metabolic process that takes place in anaerobic environments to regenerate for glycolysis, which takes place in both aerobic and anaerobic environments. Since glycolysis is unaffected by the presence of oxygen, pyruvate concentrations will be the same in either environment. During fermentation the cell changes the pyruvate into acetaldehyde. Ethanol is also a product of fermentation. We would not expect to see more acetaldehyde or ethanol in aerobic metabolism.
Adenosine triphosphate is the main product of cellular respiration, and the molecular energy of the cell. Aerobic metabolism results in a much higher yield of these energy carrying molecules due to the fact that it can use oxygen as a final electron acceptor in the electron transport chain.
Glycolysis
Glycolysis is the process that converts glucose to pyruvate. It produces a total of four ATP, but consumes two ATP, for a net yield of two ATP. Glycolysis is not dependent on the presence of oxygen and can occur in either aerobic or anaerobic environments.
Also Check: Elton John Children Biological
The Location Of Cellular Respiration
Cellular respiration takes place in both the cytosol and mitochondria of cells. Glycolysis takes place in the cytosol, whereas pyruvate oxidation, the Krebs cycle, and oxidative phosphorylation occur in the mitochondrion. Figure 1 shows the locations of the main biochemical reactions involved in cellular respiration.
The energy produced by the mitochondria is stored as potential energy in molecules called adenosine triphosphate . The main chemical produced in cellular respiration is ATP. ATP is the standard unit in which the energy released during respiration is stored. The mitochondrion can be recognized as the powerhouse of the cell because of its major role in cellular respiration. Mitochondria contain a number of enzymes to aid in this process. These organelles contain 2 membranes an outer membrane and an inner membrane. The space in between these membranes is known as the intermembrane space. The outer membrane contains many proteins known as porins and is permeable to molecules and ions . The inner membrane contains complexes involved in the electron transport chain stage of cellular respiration which will be described in more detail below.
If cellular respiration takes place in the presence of oxygen, it is known as aerobic respiration. If it takes place in the absence of oxygen, it is known as anaerobic respiration.
General Biology Questions : What Are The Three Metabolic Stages Of Cellular Respiration
Please answer the following questions: 1.- What are the three metabolic stages of Cellular respiration? 2.- How many ATP molecules are produced by Cellular Respiration and by Fermentation? 3.- Where in the cell, Glycolysis and Fermentation take place? 4.- In which cellular organelle citric acid cycle and electron transport chain take place?
2-Microscopic pores of leaf where gas exchange occurs is known as___________________________ 3-Define a Chloroplasts. 4-Photosynthesis is divided into two phases: A_______________________________________________ B_______________________________________________ 6- How many chromosomes are found in human body cells? 7-Define the Cell cycle. 8- Name the Three Phases of Interphase. 9- Name the Five stages of Mitosis. 10- Define Cytokinesis.
professional writers and delivered before your deadline
professional writers and delivered before your deadline
professional writers and delivered before your deadline
professional writers and delivered before your deadline
professional writers and delivered before your deadline
professional writers and delivered before your deadline
professional writers and delivered before your deadline
Recent Posts
You May Like: Edgenuity Algebra 1 Unit Test Answers
Aboard The Electron Transport Train
Electron transport and oxidative phosphorylation is the third and final step in aerobic cellular respiration. The electron transport chain is a series of protein complexes and electron carrier molecules found within the mitochondrial membrane in eukaryotic cells. Through a series of reactions, the “high energy” electrons generated in the citric acid cycle are passed to oxygen. In the process, a chemical and electrical gradient is formed across the inner mitochondrial membrane as hydrogen ions are pumped out of the mitochondrial matrix and into the inner membrane space. ATP is ultimately produced by oxidative phosphorylationthe process by which enzymes in the cell oxidize nutrients. The protein ATP synthase uses the energy produced by the electron transport chain for the phosphorylation of ADP to ATP. Most ATP generation occurs during the electron transport chain and oxidative phosphorylation stage of cellular respiration.
Efficiency Of Atp Production
The table below describes the reactions involved when one glucose molecule is fully oxidized into carbon dioxide. It is assumed that all the reducedcoenzymes are oxidized by the electron transport chain and used for oxidative phosphorylation.
| Step | ||
|---|---|---|
| Total yield | 30 or 32 ATP | From the complete oxidation of one glucose molecule to carbon dioxide and oxidation of all the reduced coenzymes. |
Although there is a theoretical yield of 38 ATP molecules per glucose during cellular respiration, such conditions are generally not realized because of losses such as the cost of moving pyruvate , phosphate, and ADP into the mitochondria. All are actively transported using carriers that utilize the stored energy in the proton electrochemical gradient.
- Pyruvate is taken up by a specific, low Km transporter to bring it into the mitochondrial matrix for oxidation by the pyruvate dehydrogenase complex.
- The phosphate carrier mediates the electroneutral exchange of phosphate for OHâ or symport of phosphate and protons across the inner membrane, and the driving force for moving phosphate ions into the mitochondria is the proton motive force.
- The ATP-ADP translocase is an antiporter and exchanges ADP and ATP across the inner membrane. The driving force is due to the ATP having a more negative charge than the ADP , and thus it dissipates some of the electrical component of the proton electrochemical gradient.
Read Also: What Is Mode In Math Terms
Atp Production In Cellular Respiration
How much ATP is produced in aerobic respiration? What are the products of the electron transport chain? Glycolysis provides 4 molecules of ATP per molecule of glucose however, 2 are used in the investment phase resulting in a net of 2 ATP molecules. 2 molecules of ATP are produced in the Krebs cycle. Finally, 34 molecules of ATP are produced in the electron transport chain .
How much ATP is Produced in Fermentation?
Only 2 molecules of ATP are produced in fermentation. This occurs in the glycolysis phase of respiration. Therefore, it is much less efficient than aerobic respiration it is, however, a much quicker process.
And so essentially, this is how in cellular respiration, energy is converted from glucose to ATP. And by glucose oxidation via the aerobic pathway, more ATPs are relatively produced.
Cellular Respiration As A Series Of Reduction
Redox describes all chemical reactions in which atoms have their oxidation state changed. The term comes from the two concepts of reduction and oxidation. Whereas, oxidation is the loss of electrons by a molecule or atom. An oxidation of an atom creates a cation, a positively charged ion. Just think about it being a double negative . A negative times a negative creates a positive charge.
In contrast, Reduction is the gain of electrons by a molecule or atom. By gaining electrons, it creates a positive charge on that atom or electron. A simple redox reaction is the ionic bonding of salt , where a sodium loses an electron to chlorine generating a positive charged sodium and a negatively charged chlorine. And this is how living things generate usable energy from energy stored in molecules. For every reduction, there is an oxidation. Hence, these reactions are known as redox reactions.
Read Also: Child Of Rage John Thomas
Phases Of Respiration In Organisms
Respiration occurs in the cytosol and around the plasma membrane in prokaryotic cells. In eukaryotic cells, respiration takes place in the mitochondria, which is also considered as the powerhouse of the cells.
This process is very much similar to internal combustion of the car engine, wherein organic compounds and oxygen go in, while water and carbon dioxide comes out. The energy that is liberated powers the automotive .
The three phases of Respiration are:
Example Of White Adipocytes
One of the best examples of a set of metabolic pathways that minimizes respiration occurs in white adipocytes , which are specialized to convert glucose to triglycerides . This begins with import of glucose and conversion to pyruvate in the cytosol. In the mitochondria, pyruvate is converted to oxaloacetate and Ac-CoA by pyruvate carboxykinase and pyruvate dehydrogenase. These products are condensed to form citrate, which is then exported to the cytosol for conversion to cytosolic Ac-CoA and oxaloacetate. In an ATP- and NADPH-dependent process, the adipocyte runs de novo lipogenesis to produce palmitate . The adipocyte can return three of four carbons of oxaloacetate to the mitochondria as pyruvate in a process that reoxidizes cytosolic NADH for continued glycolysis and that generates a cytosolic NADPH for continued lipogenesis . Only half of the pyruvate that enters mitochondria has to be oxidized to Ac-CoA with production of an NADH. The glucose-derived Ac-CoA is not oxidized to CO2 in the citric acid cycle, but rather is effectively exported to the cytosol to produce fat. Moreover, because the adipocyte cytoplasm can produce NADPH by running the oxidative and nonoxidative phases of the pentose phosphate pathway and by converting oxaloacetate to malate and then malate to pyruvate, it has a system to capture most of glucose’s available electrons into fat synthesis without a high oxygen demand.
Recommended Reading: What Does Cyte Mean In Biology
Results Of The Glycolysis Transition Reaction And Krebs Cycle
After glycolysis, transition reaction, and the Krebs cycle, the glucose molecule has been broken down completely. All six of its carbon atoms have combined with oxygen to form carbon dioxide. The energy from its chemical bonds has been stored in a total of 16 energy-carrier molecules. These molecules are:
- 4 ATP
- 12 NADH
- 2 FADH2
The events of cellular respiration up to this point are exergonic reactions they are releasing energy that had been stored in the bonds of the glucose molecule. This energy will be transferred to the third and final stage of cellular respiration: the Electron Transport System, which is an endergonic reaction. Using an exothermic reaction to power an endothermic reaction is known as energy coupling
Ap Biology Exam Question
Here is an example of a question about cellular respiration from the AP® Biology Exam. Lets see how you can use your knowledge to get full credit!
All of the following provide evidence of an increased rate of cellular respiration EXCEPT
increase in the concentration of CO2
a low pH in the inner membrane space
increased activity of ATPsynthase
an increase in the concentration of lactic acid
If you chose E, you are correct. As we have just learned lactic acid is a product of lactic acid fermentation, not cellular respiration.
Thank you for reading this article, Cellular Respiration: AP® Biology Crash Course Review! We really appreciate your feedback, let us know how we did!
Don’t Miss: Algebra 1 Escape Room Key
Why Do Mitochondria Have Their Own Genome
Many of the features of the mitochondrial genetic system resemble those found in bacteria. This has strengthened the theory that mitochondria are the evolutionary descendants of a bacterium that established an endosymbiotic relationship with the ancestors of eukaryotic cells early in the history of life on earth. However, many of the genes needed for mitochondrial function have since moved to the nuclear genome. The recent sequencing of the complete genome of Rickettsia prowazekii has revealed a number of genes closely related to those found in mitochondria. Perhaps rickettsias are the closest living descendants of the endosymbionts that became the mitochondria of eukaryotes.
An Overview Of Cellular Respiration
Glucose and other molecules from food are broken down to release energy in a complex series of chemical reactions that together are called cellular respiration.
Cellular respiration is a set of metabolic reactions and processes that take place in the cells of organisms. The metabolic reactions in cellular respiration allow the cell to convert biochemical energy from food into ATP. Some waste products are also produced and released from the cell . The reactions involved in respiration are catabolic reactions, which break large molecules into smaller ones, releasing energy in the process. These processes require a large number of enzymes which each perform one specific chemical reaction.
Don’t Miss: What Causes Parallax Error And How Do You Avoid It
Process Of Cellular Respiration
The overall process of cellular respiration can be likened to water flowing down a river that drives a turbine. Whereas building and maintaining the turbine are energy-dependent processes, the flow of water works with gravity so long as there is water upstream. Similarly, although producing and maintaining the mitochondrial enzymes, cell membranes, and cofactors are energy-dependent processes, fuel oxidation and respiratory electron flow are exothermic . Electrons flow in cellular respiration precisely as they flow in other electrical circuits, toward acceptors of higher electron affinity.
At the inner mitochondrial membrane, complexes I, III, and IV utilize their higher electron affinities to pump protons across the membrane to generate a proton gradient. Just as the cost of turning a water turbine is paid for by water flowing downriver, the cost of pumping protons is paid for by electrons flowing from higher-energy states to lower-energy states. See also: Proton
Return of protons through the enzyme FoF1 ATP synthase generates ATP by oxidative phosphorylation , whereas return of protons through proton pores generates heat . Because electron affinity follows a gradient pattern of O2> cytochrome C > coenzyme Q > FAD > NAD+, fuel oxidation typically transfers electrons first to NAD+ and/or FAD, and then to coenzyme Q, cytochrome C, and O2, with many additional protein-bound metal clusters as intermediates in complexes I, II, III, and IV.