Journal Of Materials Chemistry C
Materials with applications in optical, magnetic & electronic devices.
Setting up remote access to RSC content
You can find details about how to access information remotely in this step-by-step guide. The guide will also help if for any reason you have difficulty accessing the content you want.
Key Takeaways: Chemical Element Definition
- A chemical element is a substance that cannot be further broken down by any chemical reaction.
- Each element has a unique number of protons in its atom. For example, a hydrogen atom has 1 proton, while a carbon atom has 6 protons.
- Varying the number of electrons in an atom of an element produces ions. Changing the number of neutrons produces isotopes.
- There are 118 known elements.
Learning Goals Of The Major
Graduates will be able to:
Also Check: What Does I Stand For In Physics
Parent And Daughter Isotopes
When radioisotopes undergo radioactive decay, the initial isotope may be different from the resulting isotope. The initial isotope is called the parent isotope, while the atoms produced by the reaction are called daughter isotopes. More than one type of daughter isotope may result.
As an example, when U-238 decays into Th-234, the uranium atom is the parent isotope, while the thorium atom is the daughter isotope.
A Note About Stable Radioactive Isotopes
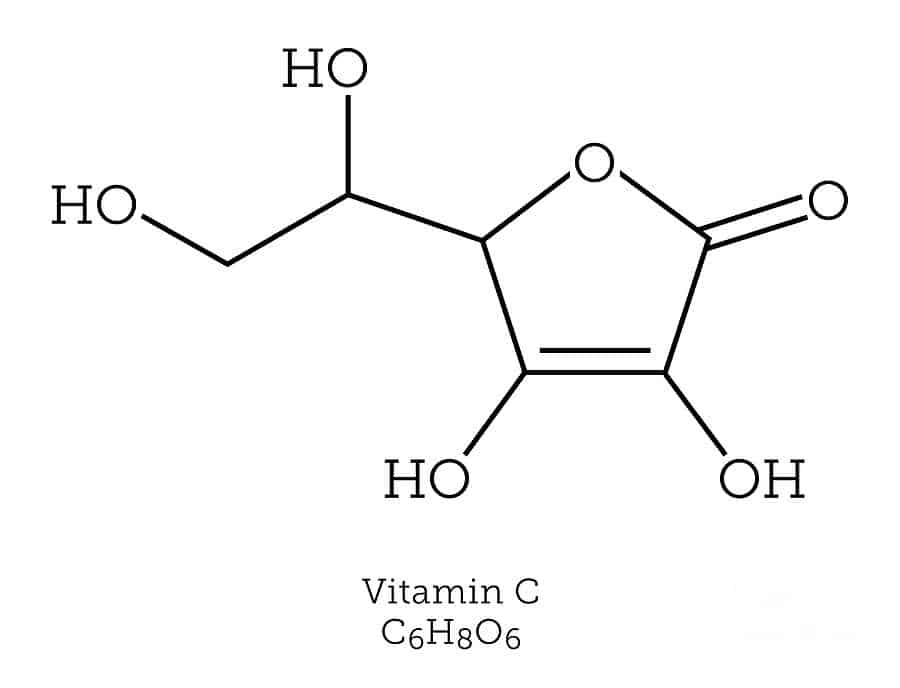
Most stable isotopes don’t undergo radioactive decay, but a few do. If an isotope undergoes radioactive decay very, very slowly, it may be termed stable. An example is bismuth-209. Bismuth-209 is a stable radioactive isotope that undergoes alpha-decay but has a half-life of 1.9 x 1019 years . Tellurium-128 undergoes beta-decay with a half-life estimated to be 7.7 x 1024 years.
Recommended Reading: What Is Plateau In Geography
Why Does Water Have A High Specific Heat
It is because of the hydrogen bonds. It fact, it is the highest S.H. of all liquids. Because water is a polar molecule, with a large electronegativity difference between hydrogen and oxygen, hydrogen bonds form between the positive hydrogen atoms in one molecule, and the negative oxygen atoms in nearby molecules. It takes a lot of energy to loosen and break the hydrogen bonds between water molecules. This is the reason that oceans cool down slower than land does.
C Meaning In Chemistry
Please also find C meaning for Chemistry in other sources.
Recommended Reading: What Is Assay In Pharmaceutical Chemistry
Chemistry Abbreviations Starting With The Letter C
- Ph.D., Biomedical Sciences, University of Tennessee at Knoxville
- B.A., Physics and Mathematics, Hastings College
Chemistry abbreviations and acronyms are common in all fields of science. This collection offers common abbreviations and acronyms beginning with the letter C used in chemistry and chemical engineering.
CADS – Chemical Agent Detection SystemCAR – Commercial And ResidentialCBRE – Chemical, Biological, Radiological ElementCBRN – Chemical, Biological, Radiological, or NuclearCC – Cubic CentimeterCCBA – Chemical Coordinate Bonding and AdsorptionCCL – Contaminant Candidate ListCDSL – Chemical Data Summary ListCDU – Chemical Dispensing UnitCFRP – Carbon Fiber Reinforced Plasticcg – CentigramCRAP – Crude Reagent And ProductsCRC – Chemical Rubber CompanyCSAC – Chemical Safety Analysis and ControlCSAD – Cysteine Sulfinic Acid DecarboxylaseCSTR – Continuously Stirred Tank ReactorCVCS – Chemical Volume Control SystemCW – Chemical Warfare
What Does C Stand For In Chemistry
Carbon
Short, what does lowercase c mean in chemistry?
Molar concentration
Beyond that, what is C stand for?
| symbol for the speed of light |
| C |
is it true, what does CS mean sexually?
The term cisgender refers to people whose gender identity and expression matches the biological sex they were assigned when they were born.
What does C stand for in math?
Complex
Read Also: What Do You Learn Psychology
See Who’s On The Team
Meet Journal of Materials Chemistry C‘s Editor-in-Chief and board members.
Natalie Stingelin, Georgia Institute of Technology, USA
Associate editors
Oana Jurchescu, Wake Forest University, USA
Monica Lira-Cantú, Catalan Institute of Nanoscience and Nanotechnology, Spain
, Imperial College London, UK
Kasper Moth-Poulsen, Institute of Materials Science of Barcelona, Spain
Neil Robertson, University of Edinburgh, UK
Federico Rosei, National Institute of Scientific Research, University of Quebec, Canada
Yana Vaynzof, Technical University of Dresden, Germany
Zhiguo Xia, South China University of Technology, China
Yadong Yin, University of California, Riverside, USA
Hao-Li Zhang, Lanzhou University, China
Ni Zhao, Chinese University of Hong Kong, Hong Kong
Zhenan Bao, Stanford University, USA
Hugo Bronstein, University of Cambridge, UK
Paola Carbone, University of Manchester, UK
Juan Casado, University of Malaga, Spain
Rajadurai Chandrasekar, University of Hyderabad, India
Lin X. Chen, Northwestern University, USA
Yen-Ju Cheng, National Chiao Tung University, Taiwan
Manish Chhowalla, University of Cambridge, UK
Chunyan Chi, National University of Singapore, Singapore
Laylay Chua, National University of Singapore, Singapore
Przemek Data, Silesian University of Technology, Poland
Olivier Dautel, University of Montpellier, France
Fernando Dias, Durham University, UK
Antonio Facchetti, Northwestern University, USA
, University of Rennes, France
Aiko Fukazawa, Kyoto University, Japan
Heat Capacity Of Water
The S.H capacity of water is associated with the unit called the calorie. In fact, one calorie is defined as the amount of heat need to raise 1 gram of liquid water one degree Celsius. The calorie was first used by Nicolas Clement in the early 19th century. It comes from the Latin word calor meaning heat.
Read Also: What Is An Experimental Study In Psychology
What Is The Difference Between Suspension And Colloid
Unlike colloid particles, those in a suspension can be separated by filtration. Colloids are capable of scattering light, but suspensions are not. Particles in a suspension can be seen with the naked eye, but those in a colloid require the use of a light microscope.
To follow more about the difference between suspension and colloid, download BYJUS The Learning App.
Put your understanding of this concept to test by answering a few MCQs. Click Start Quiz to begin!
Select the correct answer and click on the Finish buttonCheck your score and answers at the end of the quiz
Glossary Of Chemistry Terms
| Part of a series on |
|
|
This glossary of chemistry terms is a list of terms and definitions relevant to chemistry, including chemical laws, diagrams and formulae, laboratory tools, glassware, and equipment. Chemistry is a physical science concerned with the composition, structure, and properties of matter, as well as the changes it undergoes during chemical reactions it features an extensive vocabulary and a significant amount of jargon.
Note: All periodic table references refer to the IUPAC Style of the Periodic Table.
- absolute zero
- A theoretical condition concerning a system at the lowest limit of the thermodynamic temperature scale, or zero kelvins, at which the system does not emit or absorb energy . By extrapolating the ideal gas law, the internationally agreed-upon value for absolute zero has been determined as 273.15 °C .
- absorption
- 1. The physical or chemical process by which a substance in one state becomes incorporated into and retained by another substance of a different state. Absorption differs from adsorption in that the first substance permeates the entire bulk of the second substance, rather than just adhering to the surface.
- 2. The process by which matter takes up the energy of electromagnetic radiation and transforms it into any of various types of internal energy, such as thermal energy. This type of absorption is the principle on which spectrophotometry is based.
Recommended Reading: Math Caching Algebra 1 Answers
Extension Of The Symbol
In addition to the letters for the element itself, additional details may be added to the symbol as superscripts or subscripts a particular isotope, ionization, or oxidation state, or other atomic detail. A few isotopes have their own specific symbols rather than just an isotopic detail added to their element symbol.
Attached subscripts or superscripts specifying a nuclide or molecule have the following meanings and positions:
Many functional groups also have their own chemical symbol, e.g. Ph for the phenyl group, and Me for the methyl group.
A list of current, dated, as well as proposed and historical signs and symbols is included here with its signification. Also given is each element’s atomic number, atomic weight, or the atomic mass of the most stable isotope, group and period numbers on the periodic table, and etymology of the symbol.
Discovery And Recognition Of Various Elements
Ten materials familiar to various prehistoric cultures are now known to be chemical elements: Carbon, copper, gold, iron, lead, mercury, silver, sulfur, tin, and zinc. Three additional materials now accepted as elements, arsenic, antimony, and bismuth, were recognized as distinct substances prior to 1500 AD. Phosphorus, cobalt, and platinum were isolated before 1750.
Most of the remaining naturally occurring chemical elements were identified and characterized by 1900, including:
- The more common radioactive elements, including uranium, thorium, radium, and radon
Elements isolated or produced since 1900 include:
- The three remaining undiscovered regularly occurring stable natural elements: hafnium, lutetium, and rhenium
- Plutonium, which was first produced synthetically in 1940 by Glenn T. Seaborg, but is now also known from a few long-persisting natural occurrences
- The three incidentally occurring natural elements , which were all first produced synthetically but later discovered in trace amounts in certain geological samples
- Four scarce decay products of uranium or thorium , and
- Various synthetic transuranic elements, beginning with americium and curium
Recommended Reading: What Is Vector Quantity In Physics
C + O2 Is Equal To C + O How Is That Possible
C + O = CO2 . This is because carbon has valency of 4 while oxygen has valency of 2. When they react the valencies are criss crossed which means we will get C2O4 but this is simplified to CO2.
But my textbook say that C + O2 = CO2.
So how is it possible that C + O2 and C + O both get the same product of CO2?
Also you can say that the reaction of C + O = CO2 is not balanced but the balanced chemical reaction would be C + 2O = CO2 not C + O2 = CO2 which is the reaction that my textbook says.
- $\begingroup$Your question is a jumble of text, and it really is not quite clear what you’re asking right now. I suggest you make some edits to it.$\endgroup$Mar 31, 2014 at 15:11
- $\begingroup$I have explained a bit further. hope it is more clear now.$\endgroup$Mar 31, 2014 at 15:28
- 1$\begingroup$@AbhishekMhatre Only diaatomic oxygen and triatomic, ozone, exist in nature.$\endgroup$ user3735Mar 31, 2014 at 15:46
- $\begingroup$As stated Jun-Goo is not true that $C+O =CO_$ you should rearrange the question…$\endgroup$Mar 31, 2014 at 16:56
- 1$\begingroup$@Jun-Goo Kwak, because a compound it strongly reactive does not mean it does not exist. It implies that it is difficult to isolate it or store it. According to JPL Data Evaluation , there are several reactions including Atomic Oxygen .$\endgroup$
When thinking about chemical reactions it is very important to know which chemicals may react with each other. Jun-Goo Kwak already pointed out the nature of oxygen.
$$\ce$$
He assumed that:
Origin Of The Elements
Only about 4% of the total mass of the universe is made of atoms or ions, and thus represented by chemical elements. This fraction is about 15% of the total matter, with the remainder of the matter being dark matter. The nature of dark matter is unknown, but it is not composed of atoms of chemical elements because it contains no protons, neutrons, or electrons. .
The 94 naturally occurring chemical elements were produced by at least four classes of astrophysical process. Most of the hydrogen, helium and a very small quantity of lithium were produced in the first few minutes of the Big Bang. This Big Bang nucleosynthesis happened only once the other processes are ongoing. Nuclear fusion inside stars produces elements through stellar nucleosynthesis, including all elements from carbon to iron in atomic number. Elements higher in atomic number than iron, including heavy elements like uranium and plutonium, are produced by various forms of explosive nucleosynthesis in supernovae and neutron star mergers. The light elements lithium, beryllium and boron are produced mostly through cosmic ray spallation of carbon, nitrogen, and oxygen.
In addition to the 94 naturally occurring elements, several artificial elements have been produced by human nuclear physics technology. As of 2021, these experiments have produced all elements up to atomic number 118.
You May Like: What Is An Example Of Movement In Geography
Humanities And Social Sciences Breadth Requirement: Chemistry & Chemical Biology Majors
The College of Chemistrys humanities and social sciences breadth requirement promotes educational experiences that enrich and complement the technical requirements for each major.
- 15 units total includes Reading & Composition and American Cultures courses
- Remaining units must come from the following L& S breadth areas, excluding courses which only teach a skill :
Arts and Literature Philosophy and Values Social and Behavioral Sciences
To find course options for breadth, go to the Berkeley Academic Guide Class Schedule, select the term of interest, and use the ‘Breadth Requirements’ filter to select the breadth area of interest.
- Breadth courses may be taken on a Pass/No Pass basis
- AP, IB, and GCE A-level exam credit may be used to satisfy the breadth requirement
1 Elementary-level courses may not be in the student’s native language and may not be structured primarily to teach the reading of scientific literature.
2 For Chemistry and Chemical Biology majors, elementary-level foreign language courses are not accepted toward the 15 unit breadth requirement if they are used to satisfy the Foreign Language requirement.
Foreign Language Requirement
Applies to Chemistry and Chemical Biology majors only.
The LOTE requirement may be satisfied with one language other than English, in one of the following ways:
Symbols For Named Isotopes
The following is a list of isotopes of elements given in the previous tables which have been designated unique symbols. By this it is meant that a comprehensive list of current systematic symbols is not included in the list and can instead be found in the Isotope index chart. The symbols for the named isotopes of hydrogen, deuterium , and tritium are still in use today, as is thoron for radon-220 . Heavy water and other deuterated solvents are commonly used in chemistry, and it is convenient to use a single character rather than a symbol with a subscript in these cases. The practice also continues with tritium compounds. When the name of the solvent is given, a lowercase d is sometimes used. For example, d6-benzene and C6D6 can be used instead of C6.
The symbols for isotopes of elements other than hydrogen and radon are no longer in use within the scientific community. Many of these symbols were designated during the early years of radiochemistry, and several isotopes bear placeholder names using the early naming system devised by Ernest Rutherford.
| Symbol |
|---|
Don’t Miss: What Is Psychological Foundation Of Education
An Introduction To Isotopes
pangoasis / Getty Images
- Ph.D., Biomedical Sciences, University of Tennessee at Knoxville
- B.A., Physics and Mathematics, Hastings College
Isotopes are atoms with the same number of protons but differing numbers of neutrons. In other words, isotopes have different atomic weights. Isotopes are different forms of a single element.
Chemically Pure And Isotopically Pure
Chemists and nuclear scientists have different definitions of a pure element. In chemistry, a pure element means a substance whose atoms all have the same atomic number, or number of protons. Nuclear scientists, however, define a pure element as one that consists of only one stable isotope.
For example, a copper wire is 99.99% chemically pure if 99.99% of its atoms are copper, with 29 protons each. However it is not isotopically pure since ordinary copper consists of two stable isotopes, 69% 63Cu and 31% 65Cu, with different numbers of neutrons. However, a pure gold ingot would be both chemically and isotopically pure, since ordinary gold consists only of one isotope, 197Au.
Also Check: What Is Mnemonics In Psychology