Issues And Limitations Associated With Fluorescent Lipophilic Dyes
A potential problem with fluorescent lipophilic dyes is related to the fact that, because membrane dyes bind noncovalently, exposure of labeled cells to solvents that extract lipids may also extract these dyes . However, modification to many of these dyes has greatly mitigated dye loss. Nevertheless, dye retention should be tested when using tissue fixation and processing methods. Because labeling occurs by partitioning, dye concentration and cell concentration during the staining step affect the final intensity. Ideally, each laboratory should determine the optimal dye/cell ratio. Obtaining reproducible results involves greater precision of dilution and greater attention to the mixing technique than when staining with antibodies or CFSE. Lipophilic membrane dyes have some limitations that are similar to CFSE. For example, long-term tracking use is limited because dye intensity decreases in direct proportion to the number of cell divisions. Their high-labeling intensity can complicate filter selection and color compensation when other probes are used. Additionally, monitoring of proliferation by dye dilution is affected by artifacts resulting from the presence of apoptotic and necrotic cells.
Synthesis And Application Of Quantum Dots
Quantum dots must first be synthesized and their surface modified so that they are biologically compatible. Generally, a quantum dot has a three-layer structure composed of a core, shell and polymer coatings. The most common quantum dots used have a CdSe core that is usually coated with a zinc sulfide shell to improve photoluminescence of the quantum dots. Polymer coatings that surround the shell are used to make the materials soluble to water. The surface of quantum dots should be further modified so that the dots can be directed to a target. There are three main methods to target a biocompatible quantum dot: using antibodies with peptides and using small molecules. Each of these approaches has advantages and disadvantages, and none of the approaches is universal for all applications.
Quantum dots can be used for specific labeling of individual cell surface biomolecules, as well as for targeting intracellular single biomolecules, such as mRNA and molecular motors. The application of quantum dots is similar to that of organic fluorophores. After a ligand is attached to the label, this conjugate binds to its target receptor, which is then viewed by the fluorescence of the label using an optical microscope. For labeling only a few cells, fluorescence-labeled ligands can be injected into live cells using micropipettes. Electroporation can also be used .
Probes For Protein Catabolism
The nematode worm C. elegans is a premier system for dissecting the genetic control of longevity and age-related pathologies including protein homeostasis and folding. However, the available tools for studying the induction of autophagy and autophagic flux in worm are rather limited. In collaboration with the Miller Lab are working to develop sensitive, selective, cell lineage-specific probes for autophagy in the worm.
Also Check: Who Are Paris Jackson’s Biological Parents
Main Difference Probe Vs Primer
PCR is a technique used in biotechnology to amplify specific DNA fragments for various purposes. Probe and primer are two types of single-stranded, oligonucleotides used in various types of PCR. Probes are mainly used in qPCR while synthetic primers are used in every type of PCR. The main difference between probe and primer is that probe is that probe is used to detect the presence of a specific DNA fragment in the mixture through the hybridization with a double-stranded DNA whereas primer is used in the initiation of the polymerase chain reaction by hybridization with single-stranded DNA. Generally, primers are used in the initiation of DNA replication inside the cell. Probes are used in hybridization reactions as well.
Summary Probe Vs Primer
Probe is a small fragment of DNA or RNA sequence that can be hybridized with complementary nucleotides to detect a target sequence in the sample. Probes are labeled radioactively, immunologically or fluorescently to see the presence of target sequence. Primer is a very small DNA or RNA fragment that act as the starting point for in vitro DNA amplification. DNA polymerase identifies 3 OH group primer and initiates the building of new strand complementary to the template. Probes and primers work similarly by hybridizing with complementary nucleotides. Thus, the key difference between probe and primer is their prime function.
Reference:1.Primer . Wikipedia. Wikimedia Foundation, 02 Feb. 2017. Web. 01 Mar. 2017.2.Hybridization probe. Wikipedia. Wikimedia Foundation, 30 Dec. 2016. Web. 01 Mar. 20173.Primer . Primer ScienceDirect Topics. N.p., n.d. Web. 01 Mar. 2017
Image Courtesy:1. FISH for Bacterial Pathogen Identification By Pepetps Togopic Ivan Akira Magnus ManskeTimothy W. Ford via Commons Wikimedia2. Primers RevComp Melted2 By Richard Wheeler Own work via Commons Wikimedia
Read Also: Geometry Segment Addition Postulate Worksheet
Biotinylation Of Dna And Oligonucleotide Probes
DNA probes can be labeled with biotin for subsequent detection with streptavidin-labeled alkaline phosphatase by a number of enzymatic and chemical methods. Biotin can be incorporated into DNA probes enzymatically by replacing one nucleotide with a biotinylated nucleotide derivative. Biotinylation does not significantly affect the hybridization conditions of the probe. Several biotinylated dUTP analogs and other biotinylated nucleotides are available from commercial suppliers.
Synthetic oligonucleotide probes can be 3 end-labeled using terminal transferase, allowing one or more biotinylated nucleotides to be added.21We have found that optimum sensitivity is obtained when 10 or fewer biotinylated nucleotides, spaced with unlabeled nucleotides at a ratio of 1:3, are added to the 3 end of an oligonucleotide probe. Spacing of biotinylated nucleotides with unlabeled nucleotides permits more efficient binding of multiple streptavidin-labeled alkaline phosphatases to the hybridized probe.
Cloned DNA probes can be labeled with biotin by using biotinylated nucleotides in nick translation2 and random hexamer priming23 reactions. For both of these systems, we replace dTTP with biotin-11dUTP in standard labeling reactions. These labeling systems produce DNA probes with high levels of biotinylation that can be used in a variety of blot detection systems.
Louise O’Connor, Majella Maher, in, 1999
Primary Antibodies As Probes
Thousands of primary antibodies are commercially available for protein targets with a history of investigation in biological research. Except for a few very popular research targets, these primary antibodies are offered without detectable tags, and some sort of secondary detection method is required. Nevertheless, nearly any antibody can be labeled with biotin, HRP enzyme or one of several fluorophores if needed.
Depending on the application to be performed, different levels of purity and types of specificity are needed in a supplied primary antibody. To name just a few parameters, antibodies may be monoclonal or polyclonal, supplied as antiserum or affinity-purified solution, and validated for native protein or denatured protein detection.
If no antibodies exist for an antigen of interest, new antibodies can be produced using well established techniques for immunizing animals with prepared forms of the antigen. A variety of reagents are available to assist in antibody production and purification, and various companies specialize in antibody production services.
Recommended Reading: My.hrw Algebra 1
Hybridization Probes Definition Uses Preparation Types
THIS BLOG INCLUDES: hide 1 Hybridization Probes Definition 2 Preparation of Hybridization Probes 3 Types of Hybridization Probes 4 Labeling of Hybridization…
Sourav BioThis article writter by Sourav Bio on June 18, 2020
Writer and Founder of Microbiologynote.com. I am from India and my main purpose is to provide you a strong understanding of Microbiology.
Class Ii Odorant Receptors
In contrast to class I, class II OR genes account for a large share of the mammalian OR repertoire. ISH analyses with various OR gene probes demonstrated that each OR gene is expressed in a restricted area in the OE. It has been reported that OSNs expressing a given OR gene are confined to one of the four OE zones but are randomly distributed within the zone . This appears to be true for zone 1-specific class I genes . However, for most class II OR gene, the expression area is not always confined to one of the four conventional zones . Expression areas appear to be specific to each OR gene and are arranged in an overlapping and continuous manner in the OE. It was demonstrated by DiI retrograde staining experiments that the dorsal/ventral arrangement of glomeruli in the olfactory bulb is well correlated with the expression areas of corresponding ORs along the dorsomedial/ventrolateral axis in the OE . How is the area-specific expression regulated for the OR genes? It has been assumed that zone-specific transcription factors for the OR genes are responsible for the regulation. However, if the expression areas of various ORs are arranged in a continuous and overlapping manner, we may have to consider an alternative mechanism, one that detects a gradient or the relative location in the OE for each OR gene along the DM/VL axis. If this is the case, the choice of the OR genes may be more restricted by the location of the OSN in the OE than what has been thought.
Read Also: Eoc Fsa Practice Test Algebra 1
What Is A Probe
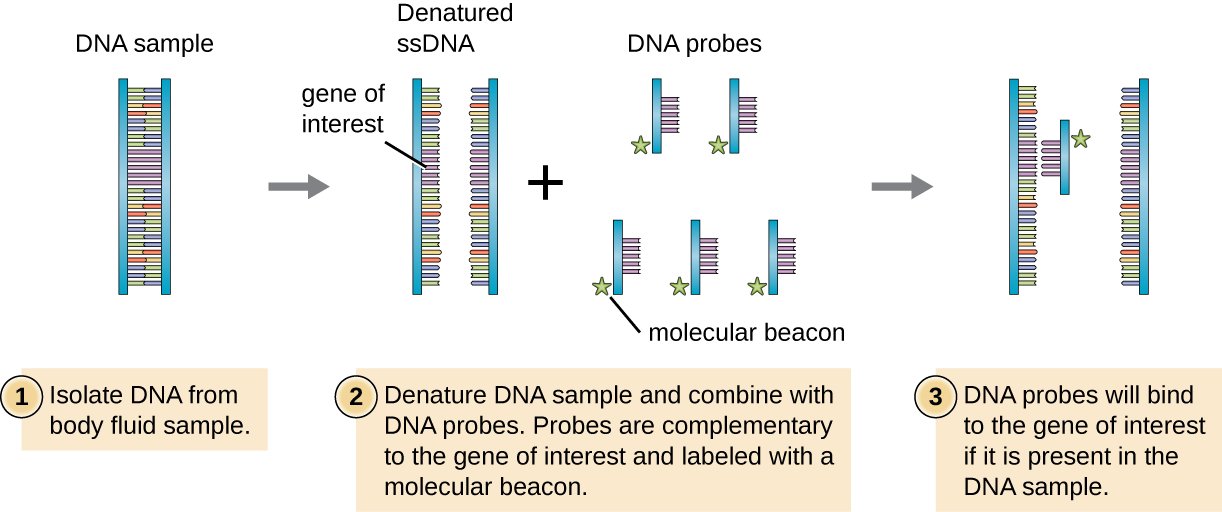
Probe is a fragment of DNA or RNA used to detect the presence of a specific DNA fragment within a sample. Therefore, probes can be used for two types of techniques, in qPCR and in hybridization reactions. Four facts should be considered in the designing of a probe.
In qPCR, probes are labeled with fluorescent dyes or radioactive elements. These probes are hybridized with the target sequence in the DNA duplex. Different types of labeled probes, either with radioactive elements or fluorescence, are used in various types of hybridization reactions as well. The hybridization of the PNA probes to their target sequences are shown in figure 1. PNA probes are used to determine the length of the telomeres.
Figure 1: Hybridization of PNA Probes
During hybridization, probes bind to the single-stranded DNA in a complementary manner.
Nucleic Acid Hybridization/annealing And Stringency
Hybridization or annealing is based on the ability of a single-stranded nucleic acid sequence to specifically bind to its/a complementary strand. If dsDNA is denatured or melted, it will hybridize when the denaturant or heat is removed. A few bases start the process and the rest of a matching sequence will hybridize much like a zipper closing. Hybridization in molecular methods uses single-stranded nucleic acid probes or oligonucleotides of defined sequence to hybridize to a target DNA or RNA of interest. Complementarity between the two strands is determined by the formation of specific hydrogen bonds between nucleotide bases of the probe/primer and target nucleic acid, such that only the base pairs adenine-thymine, adenine-uracil, and guanine-cytosine form hydrogen bonds, giving sequence specificity to the double stranded duplex.
Recommended Reading: How To Calculate Ihd Organic Chemistry
Live Imaging Of Transcription Using Active Rna Polymerase Ii
by Tokyo Institute of Technology
A genetically encoded “live-cell” probe designed by researchers from Tokyo Tech has been able to identify phosphorylated Ser2 in the enzyme RNA polymerase II, subsequently identifying sites of the elongation phase of active transcription in living cells. This probe could be used to specifically image transcription elongation in real time in living animals, furthering gene regulation research by leaps and bounds.
In nuclei of all eukaryotic cells, an enzyme known as RNA polymerase II is responsible for transcribing nearly all the genes that give us our different characteristics. Therefore, to get a better idea of the entire eukaryotic genome regulation and the functions of various cells, RNAP2 behavior in live cells has been analyzed using fluorescent protein-tagged RNAP2 molecules. While this approach has advanced our knowledge on this protein’s behavior significantly, a cell typically contains more than 100,000 RNAP2 molecules and thousands of active transcription sites or “factories,” and it is very difficult to track single RNAP2 foci when the entire cell lights up like a lightbulb because of the fluorescence!
What does this mean for the future of genetic research? “Understanding the regulation of RNAP2 can help us understand genomes, cell functioning, and phenotypes,” Prof. Kimura elaborates.
The promising results of this study could fuel future research in genetics and gene regulation and widen our knowledge about the human genome.
Preparation Of Hybridization Probes
A Hybridization Probe can be prepared by various techniques
- Highly purified mRNA can be used as a probe.
- Single-stranded RNA probe can be prepared by cloning the cross ponding DNA sequence into a special vector.
- Single-stranded cDNA probe can be prepared by limiting the copying of mRNA by reverse transcriptase to only one stand.
Also Check: Geometry Unit 4 Test Answer Key
Fluorescent Probes For Biological Imaging
Xuanjun Zhang
1Faculty of Health Sciences, University of Macau, Taipa, Macau
2Department of Chemistry, Anhui University, Hefei 230601, China
3Department of Chemistry, University of Washington, Seattle, WA 98195-1700, USA
Owing to the high sensitivity, high resolution, and the wealth of contrast mechanisms, fluorescence imaging is the most versatile and widely used visualization modality to study the structure and function of biological systems and the molecular process in living organisms without perturbing them.
Fluorescence molecular imaging is an evolving field of imaging sciences, which involves the development of microscopic techniques for live cell imaging at super resolution and macroscopic techniques to monitor molecular events in living organism. The breakthrough of super-resolution techniques allows researchers to obtain fluorescence images with a higher resolution than the diffraction limit. On the other hand, fluorescence imaging is also facing important challenges. Because the imaging requires exogenous probes to enhance imaging contrast or provide signal readout, the probe performance largely determines the detection limit and sensitivity. The intrinsic poor penetration of UV and visible light limits their broad applications in biology. Therefore, promising probes that exhibit high photostability, long fluorescence lifetime, strong absorption, and/or emission in the near-infrared region are highly desirable.
Acknowledgments
Xuanjun Zhang
Key Difference Probe Vs Primer
The molecular probe is a small DNA or RNA fragment that recognizes the complementary sequences in DNA or RNA and allows identification of the target sequence. Primer is a small stretch of DNA or RNA which serves as a starting point for DNA synthesis. Primers and probes hybridize with the complementary nucleotides of the template DNA or the target DNA. However, the key difference between probe and primer is that primers are necessary for DNA replication while probes are necessary for detection of specific sequences in the sample DNA.
CONTENTS5. Summary
Also Check: What Does Abiotic Mean In Biology
Issues And Limitations With Quantum Dots
It is important to minimize nonspecific binding with quantum dots because this can lead to the misinterpretation of experimental results. Rigorous controls must also be performed in quantum dot experiments . The largest problem associated with quantum dots is toxicity . Because the nanomaterials of quantum dots are composed of heavy metals, they are potentially toxic during in vitro and in vivo imaging. Quantum dot toxicity depends on many parameters, such as size, shape, composition, charge, redox activity, concentration, surface coating, photostability, solubility, and exposure time. Toxicity is generally caused by two sources: the composition of the semiconductor materials, especially the core composed of heavy metals and the generation of free radicals during excitation. The toxicity of quantum dots to cells and animals is time and dose dependent. Therefore, when imaging with a low concentration of quantum dots within a certain time, they can be ideal probes. Another issue with quantum dots is that the addition of specialized coatings means that the final tracking agent is larger than GFP or streptavidin and much larger than small organic dyes such as CFSE and PKH26 . Therefore, quantum dots are unable to enter cells unless taken up by naturally occurring internalization mechanisms or coupled to targeting molecules that assist in cellular uptake.
Ahybrid Stability As The Basis Of Dna Probing
DNA probes and sample DNAs or RNAs are capable of hybridizing each other through the complementary base-pairing and result in double-stranded hybrid formation. The strength of DNA probe hybridization to sample molecules is determined by the stability of the hybrid duplex formed. The other parts of each sample molecule, which is usually longer than DNA probe, remains on either side of the hybrid duplex and does not affect the hybridization strength since they have no interactions with the probe.
The formed hybrid duplexes may be melted into single-stranded random coils by elevating temperature. Their thermal stability under a given solvent condition is determined by three factors: the G+C content and base sequence of DNA probes, the length of hybrid duplexes, and base mismatches in hybrid duplexes. The last factor allows the detection of base sequence difference between DNA probes and sample DNAs or RNAs using the thermal stability of the hybrid duplex, and thus provides the basis of DNA probing.
J.M. WagesJr, in, 2005
Recommended Reading: Unit 1 Test Geometry Basics Answers Key
Uses Or Application Of Hybridization Probes
Fluorescent Probes For The Simultaneous Detection Of Multiple Analytes In Biology
E-mail:
b Institute of Bio-organic Chemistry, Polish Academy of Sciences, Poznan, Poland
c State Key Laboratory of Applied Microbiology Southern China, Guangdong Provincial Key Laboratory of Microbial Culture Collection and Application, Guangdong Institute of Microbiology, Guangdong, People’s Republic of China
You May Like: Renate Blauel 2013