What Does Inert Mean In Chemistry
Definition. An inert chemical substance is one that is not generally reactive. This is a synonym for inactive with respect to chemical reactions. Inert has a non-chemical meaning of being unable to move or resist movement for example, the accident victim was laying on the ground, inert.
Does adding an inert gas increase pressure?
Add an inert gas to the constant-volume reaction mixture: This will increase the total pressure of the system, but will have no effect on the equilibrium condition. That is, there will be no effect on the concentrations or the partial pressures of reactants or products.
Definition Of Inert Chemistry Dictionary Chemicool
- www.chemicool.com
- Highest rating: 4
- Summary: An inert chemical is one that is stable and unreactive under specified conditions. At one time, the noble gases in Group 18 of the periodic table were known
See Details
- Highest rating: 4
- Summary: Chemistry. having little or no ability to react, as nitrogen that occurs uncombined in the atmosphere. Pharmacology. having
See Details
Which Is Inert And Which Is Labile In D7metal
Low spin complexes of d7metal ions are also found to be labile due to CFSE gain. It can be seen that d4low spin are also labile in nature. On the other side, d3and d8metal complexes are inert in nature and undergo slow ligand displacement through the associative pathway.
Whats the difference between labile and inert bonds?
Lability refers to how easily metal-ligand bonds are broken. A compound in which metal-ligand bonds are easily broken is referred to as labile. A compound in which metal-ligand bonds are more difficult to break is referred to as inert.
Read Also: Mcdougal Littell Algebra 2 Chapter 2 Test Answers
Safeopedia Explains Inert Gas
The term inert gas is somewhat of a misnomer, as these gases can, in fact, be reactive under certain conditions. As such, in the context of chemistry and materials science, these gases are generally referred to as noble gases. The term noble has historically been used in chemistry to describe the reluctance of certain metals to react chemically, and the term noble gas is used to connote that same reluctance.
Can You Define An Inert Chemical
Alan Newport| Nov 29, 2017
A current flap in my own community reminds me how important it is to understand the meaning of words and how they are used in our complex society.
The background to my story is that a group of several tribes leased an old Indian boarding school, known as Chilocco, to the Department of Homeland Security. The Indian school closed in 1980. Now the DHS has announced it will be testing the movement of biological weapons using “inert” chemicals among some of the buildings. I live nearby. My hometown of Newkirk, Oklahoma, is about seven miles south. The town of Arkansas City, Kansas, is about four miles north.
At last count I saw reported, more than 8,500 residents from the area had signed a petition against the testing and most, if not all, the tribes are upset and/or protesting.
The deception and the lingual treachery about this event, to my mind, is the DHS claim that the testing is safe because these are “inert” chemicals.
That is a prima facie lie.
Those of us who labor in the agricultural industry should know as well or better than anyone that the claim of a chemical ingredient being “inert” is an explanation that chemical does not have direct effect on the purpose and label claims of the product.
Everything you might need to know about the properties of inert ingredients is available on the Environmental Protection Agency websites, but for your immediate edification I’ll list a few important facts here.
Read Also: What Is Ug In Physics
Examples Of Inert In A Sentence
inertinertinertForbesinert alinertThe Atlanticinert The Weekinert San Francisco ChronicleinertCNNinertPopular Mechanicsinert Robb Report
These example sentences are selected automatically from various online news sources to reflect current usage of the word ‘inert.’ Views expressed in the examples do not represent the opinion of Merriam-Webster or its editors. Send us feedback.
What Is An Inert Surface 3 Ways To Tell If A Flowpath Is Inert
If you’re familiar with the SilcoTek® website and blogs you know that we offer inert coatings. But what does that mean? A web search will tell you that an inert substance is not chemically reactive, has no ability to react, or does not produce a chemical reaction. That sounds suspiciously absolute to me.
I guess I inherited some of my mother’s skepticism. Like when I told my mom I absolutely did not try to burn down the garage. At which point my mother looked down her glasses at me and said “really, you’re absolutely sure you had nothing to do with trying to burn down the garage. You had nothing to do with the candle, the batteries, absolutely nothing to do with it.” At which point the nuance of the situation made me wonder if I absolutely had anything to do with the “incident”. So I fessed up that I may have had something to do with it. After all what in life is ever absolute? As with that “incident” which I will never discuss again the absolute statement of being non reactive or no ability to react seems a bit fishy.
So what is an “inert surface” and how do I know if I have one?
Digging further into the definition you’ll find the term gets less clear. You’ll find that given enough energy and under the right conditions almost anything will react. It’s all about the conditions and specific chemicals under which the surface should be non reactive. So be weary of claims of absolute inertness.
Here are 3 tips to determine if a surface is reacting with your sample.
Read Also: How To Learn Chemistry Easily
Can We Eat Nitrogen
FDA says that whileliquid nitrogen is nontoxic, it can cause severe damage to the skin and internal organs if mishandled or accidently ingested due to the extremely low temperatures it can maintain. There is also the risk of breathing problems, which may be especially problematic for those that suffer from asthma.
What Is Helium Inert
helium , chemical element, inert gas of Group 18 of the periodic table. The second lightest element , helium is a colourless, odourless, and tasteless gas that becomes liquid at 268.9 °C .
What are chemical properties of helium?
Helium is a colourless, odourless, insipid and non-toxic gas. Its less soluble in water than any other gas.Chemical properties of helium Health effects of helium.
| Atomic number |
|---|
| 268.9 °C |
Recommended Reading: How To Find The Sum In Math
How Does An Inert Electrode Work
Inert electrode is a metal that does not take part or interfere in any chemical reaction. But it is still used to conduct electricity by transferring electrons with the solution rather than exchanging ions with the solution. Inert electrode may supply or withdraw electrons in the process of conducting electricity.
Is oxygen reactive or inert?
Element No. 8 on the Periodic Table of the Elements is a colorless gas that makes up 21 percent of Earths atmosphere. Because its all around, oxygen is easy to dismiss as dull and inert in fact, its the most reactive of the non-metallic elements.
How Do You Use Inert
Inert in a Sentence
What is the difference between inert and inert gas?
Both are filled with an inert gas. An inert gas is a gas that is generally non-reactive with other substances. The term inert means non-reactive. We refer to gases as being chemically inert if their atoms dont combine with other atoms in chemical reactions.
Which of the following gases is called inert gases?
Nitrogen gas and carbon dioxide are referred to as inert gases because of their very low reactivity. These gases are not inert in the same way as the noble gases, which exist in their elemental form. They do behave similarly to the noble gases, however.
What are the uses of inert gases in everyday life?
Inert gases have many practical uses because of their low reactivity. They are often mixed with non-inert gases to decrease the reactions of the non-inert gases with substances. Many are used in lighting, and some have important uses in nuclear reactors and in medicine.
Don’t Miss: What Is Endpoint In Chemistry
What Is The Mechanism Of Inert Atmosphere Reactions
Inert atmosphere reactions. Afterward, the needle penetrates the septum that is used as cap of the container in which the reaction takes place, and the puncture of a second needle prevents overpressure. Nitrogen displaces the air and keeps the inside of the container free of oxygen. Another option, which is also used at the miniscale,
What Happens When You Put Helium And Sodium Together
They found that if you put sodium and helium together and compress it to pressures like at the center of the Earth, sodium can actually react with helium and form stable compounds. At first some scientists thought helium might be sharing electrons after all.
What are the chemical properties of helium gas?
The Chemical Properties of Helium are Tabulated Below. Humans have no sense that they can detect the presence of Helium. Although Helium is non-toxic and inert, it can act as a simple asphyxiant by displacing oxygen in the air to the below-required levels to support life.
Read Also: What Is Longshore Drift In Geography
What Is Inert Pair Effect In Chemistry
The inability of participation of valence shell s-orbital electrons in the chemical reactions of the element and their inability to ionize is called the inert-pair effect. Why do B and Al not show the inert-pair effect?
What is inert electron?
Inert electron essentially means that it is reluctant to participate in a chemical reaction. This reluctance can have causes, but some important ones are inert-pair effect, poor shielding effect, and pairing between electrons of opposite spin.
Is Fe3+ Kinetically Inert
d3 Cr3+ ion is inert and the water molecules do not dissociate as quickly as the basic ligand deprotonates the acidic coordinated water molecules. 25.
What is the difference between labile and inert complexes?
What does kinetically inert mean?
Kinetically inert means that the activation energy required for product formation is very high. Be and Mg have very less reduction potential E0 compared to other members of the same group. This indicates that they are less electropositive, hence do not reacts with cold water.
How are inert complexes different from labile complexes?
The key difference between inert and labile complexes is that inert complexes undergo slow substitution, whereas labile complexes undergo rapid substitution. This is mainly because inert complexes are thermodynamically stable complexes with a large activation energy barrier.
Also Check: What Is Kinetic Energy In Physics
Why Are Helium Atoms Stable And Inert
This is because a helium atom is loath to give up its two electrons, which perfectly fill its only electron shell. Elements with shells that are already full and have no electrons to lend are called noble gasesand helium, the smallest of these, is considered the most inert.
How does helium relate to chemistry?
What Is Nitrogen In Coffee
Typically made from cold brew coffee, nitro coffee is a beverage that has been infused with nitrogen gas, which gives the drink its name. Nitrogen gas, which is also used in varieties of beer like porters and stouts, creates small bubbles in the coffee, and gives the drink a thick and creamy texture.
Also Check: What Is How Many In Math
What Does Labile Mean In Chemistry
Labile is an adjective used to describe something that is easily or frequently changed. For example, in chemistry, a compound that can be easily broken down by heat is called labile. The term can also be used in psychology to describe someone who is emotionally unstable.
What are the difference between labile and inert complexes?
The key difference between inert and labile complexes is that inert complexes undergo slow substitution, whereas labile complexes undergo rapid substitution. Labile complexes, on the other hand, are thermodynamically unstable, and they have a very small activation energy barrier.
What Does It Mean When Something Is Inert
Definition of inert 1 : lacking the power to move. 2 : very slow to move or act : sluggish. 3 : deficient in active properties especially : lacking a usual or anticipated chemical or biological action.
What is the difference between a reactive and inert element?
In context|chemistry|lang=en terms the difference between reactive and inert. is that reactive is that readily takes part in reactions while inert is a substance that does not react chemically.
What are inert reactions?
An inert gas is a gas that does not undergo chemical reactions under a set of given conditions. The noble gases often do not react with many substances and were historically referred to as the inert gases. Unlike noble gases, an inert gas is not necessarily elemental and is often a compound gas.
What is an example of inert?
The definition of inert is slow or having no action or power to move. An example of inert is a slug that is standing still.
Read Also: Bju Algebra 1 3rd Edition
Where Can Helium Be Found Under Inert Gas
Helium falls under inert gas since its outermost electron orbital is full with two electrons. Helium can also be found in lasers, compressed air tanks and coolant in nuclear reactors. It holds the lowest boiling and melting points among the all other elements.
Which is the correct atomic number for helium?
Safety Summary: Laboratory Chemical Safety Summary Helium is a noble gas with the atomic symbol He, atomic number 2, and atomic weight 4.003. It is a colorless, odorless, tasteless gas that is not combustible and does not support combustion.
What Do You Mean By Inertness Of Nitrogen
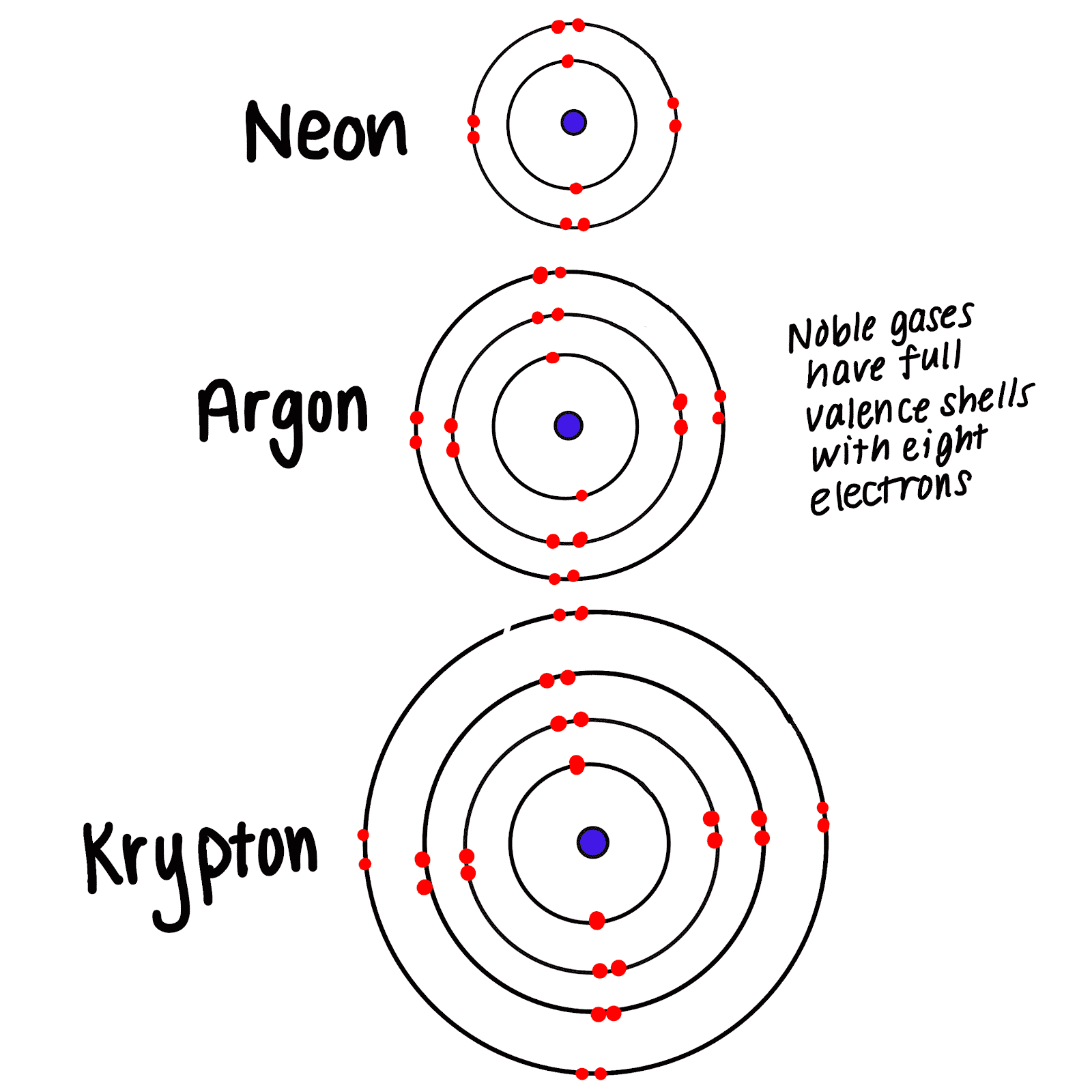
What do you mean by inert gas?An inert gas is a gas which does not undergo chemical reactions under a set of given conditions. The noble gases often do not react with many substances, and were historically referred to as the inert gases.example argon,neon
Why is nitrogen called an inert gas?
The term inert gas refers to non-reactive gas. It is also termed a noble gas. Nitrogen is called an inert gas, because of its physical appearance. It is a colourless, tasteless and odourless gas.
Why nitrogen is an inert element?
Solution : Nitrogen is inert because its `p`-subshell is half-filled due to it has maximum exchange energy. Thus it is exists in stable form as like inert gas and it form triple bond `.
What are inert gases Class 9?
There are some elements which do not combine with other elements. These elements are : Helium,Neon, Argon, Krypton, Xenon and Radon. They are known as noble gases or inert gases because they do not react with other elements to form compounds.
Don’t Miss: What Is Variation In Math
What Are Inert Complexes
Inert Complex is the transition metal complex ion that proceeds slowly or not at all and requires a large activation energy to the ligand substitution, where as complexes proceed quickly reached equilibrium with ligands are the Labile Complex .
Does labile mean reactive?
What is Labile? The term has loosely been used to describe a relatively unstable and transient chemical species or a relatively stable but reactive species. It must therefore not be used without explanation of the intended meaning. See also inert , persistent , reactive , unreactive.
Is Co3+ inert kinetically?
In summary, we have demonstrated that Co3+ can mediate the interaction between the His6-tag and NTA and forms a complex that is inert towards ligand exchange, is thermo- dynamically stable, and does not react with chelators or reducing agents.
What do you mean by labile?
1 : readily or continually undergoing chemical, physical, or biological change or breakdown : unstable a labile mineral. 2 : readily open to change has so labile a face that some of her scenes rock with emotion Manny Farber.
Properties Of The Halogens
Also Check: How To Improve Your Psychological Health
Stability Of A Plastic
A plastic is heated in an inert atmosphere or in oxygen or air until it is completely decomposed and the changes connected with the thermal degradation process are recorded by thermogravimetry or DSC. Often, differential thermogravimetry curves are characteristic of certain plastics and can, therefore, be used for identification purposes. The behavior of plastics during oxidation is important for their practical use and for their recycling or environmental decomposition. The method permits the determination of melting temperature Tm, crystallinity, and the oxidation induction temperature as shown in Figure 4.
Figure 4. Thermal analysis of polypropylene and polyethylene . Crystallinities, melting temperatures, Tm, and oxidation induction temperatures, To, are indicated. Thermal analysis of plastics. American Laboratory 19: 98103.)
Shaowei Chen, in, 2007
What Does Non Inert Mean
Not inert chemically active. Natural materials, or those having the lowest content of non-inert substances or least emission of volatile or gaseous components, are selected.
Does the word inert mean active?
having no inherent power of action, motion, or resistance : inert matter.
Why inert gases are stable?
They are the most stable due to having the maximum number of valence electrons their outer shell can hold. Therefore, they rarely react with other elements since they are already stable.
What is the meaning of inert atmosphere?
The term inert means chemically inactive, so an inert atmosphere is an environment in which powder bed fusion can take place without the risk of contamination from reactive gases that exist in the air, such as oxygen and carbon dioxide.
Read Also: What Is Endocytosis In Biology