Evaluating The Chemical Stability Of Metal Oxides In So3 And Applications Of Sio2
Department of Chemical Engineering, Graduate School of Engineering, Hiroshima University, Higashi-Hiroshima, Japan
Correspondence
Toshinori Tsuru, Department of Chemical Engineering, Graduate School of Engineering, Hiroshima University, 1-4-1 Kagamiyama, Higashi-hiroshima 739-8527, Japan.
Department of Chemical Engineering, Graduate School of Engineering, Hiroshima University, Higashi-Hiroshima, Japan
Correspondence
Toshinori Tsuru, Department of Chemical Engineering, Graduate School of Engineering, Hiroshima University, 1-4-1 Kagamiyama, Higashi-hiroshima 739-8527, Japan.
Hazards Of Sulfur Trioxide
When inhaled, remove the victims to fresh air immediately and check for breathing, then get medical assistance. When ingested first rinse the mouth thoroughly with water and this water must not be swallowed, do not induce vomiting, then give the victim milk or water, the quantity depends upon their age. If the eye is exposed to it then rinse it thoroughly with lukewarm water for a minimum of 15 min.;
Setting Your Browser To Accept Cookies
There are many reasons why a cookie could not be set correctly. Below are the most common reasons:
- You have cookies disabled in your browser. You need to reset your browser to accept cookies or to ask you if you want to accept cookies.
- Your browser asks you whether you want to accept cookies and you declined. To accept cookies from this site, use the Back button and accept the cookie.
- Your browser does not support cookies. Try a different browser if you suspect this.
- The date on your computer is in the past. If your computer’s clock shows a date before 1 Jan 1970, the browser will automatically forget the cookie. To fix this, set the correct time and date on your computer.
- You have installed an application that monitors or blocks cookies from being set. You must disable the application while logging in or check with your system administrator.
Read Also: Sacred Geometry Moon Phases Tattoo Spine
Properties Of Sulphur Dioxide
- SO2 dissolves in water to form sulphurous acid due to which it possesses an acidic character.
H2O + SO2; H2SO3
- It does not support combustion nor is it combustible.
- SO2 is a strong oxidizing agent.
2H2S + SO2; 3S + 2H2O
- It also acts as a reducing agent.
SO2 + 2H2O H2SO4 + 2H
- In the food industries, as a food preservative.
- It acts as a bleaching agent to remove the excess chlorine and as a disinfectant.
- In cold storage plant, it acts as a refrigerant.
- It is used as a reagent;and a solvent in the laboratory.
Safety measures have to be taken against sulphur dioxide as inhaling this gas causes serious health problems such as premature death and respiratory problems.
What Is The Difference Between So2 And So3
SO2 is sulfur dioxide while SO3 is sulfur trioxide. Both are oxides of sulfur. The key difference between SO2 and SO3 is that SO2 is a colourless gas at room temperature, whereas SO3 is a colourless to white crystalline solid. Furthermore, the oxidation state of sulfur in sulfur dioxide is +4 while in sulfur trioxide it is +6. Due to the presence of a lone electron pair and their geometry, sulfur dioxide is a polar compound, while sulfur trioxide is a nonpolar compound. The following infographic summarizes the;difference between SO2 and SO3.
Don’t Miss: Eoc Fsa Warm Ups Algebra 1 Answers
Our Expert Chemistry Tutor Sabrina Took 2 Minutes And 9 Seconds To Solve This Problem
So3 chemistry name. Being an intelligent and well-practiced human being you must know what is molecular geometry but let me revise it for the all young students out thereMolecular geometry is the three-dimensional structure of the atoms which helps in the constitution of a molecule. Search for Other Answers. A staff officer of the third class usually a junior officer.
How long does this problem take to solve. The name of the molecular compound of chemical formula SO3 is As a gas this is a pollutant agent of acid rain. Sulfur trioxide a chemical compound of sulfur.
The chemical equation is given below. Click here for details. The chemical name for SO3 would be sulfur trioxide.
Sulfur trioxide reacts with a base sodium hydroxide forms sodium hydrogen phosphate. SO3 may refer to. WEB SEARCH MSDS.
ClO2 chlorite ion. OTHER SETS BY THIS CREATOR. It is used as a precursor to sulfuric acid.
It is corrosive to metals and tissue. NO3 nitrate ion. Sulfur Trioxide Molecular Geometry.
The common names and formulas of chemical compounds is a very relevant area of study for every competitive exam including SSC and it is important for the aspirants to develop a fair idea on this subject. Even hypothetical chemical compounds need to have correct names. Often shipped with inhibitor to prevent polymerization.
Try a smart search to find answers to similar questions. General Chem Common Cations 25 Terms. Search by Systematic name Synonym Trade name Registry number SMILES or InChI.
What Is The Ph Of Sulfur Dioxide
In juices with high acidity less sulfur dioxide is required, for example 15 mg l1 with free sulfur dioxide at pH 3.0 has the same antimicrobial effect as 150 mg l1 at pH 4.0 Percentage of sulfite, bisulfite and molecular sulfur dioxide in aqueous solution as a function of the pH.
Get in touch with BYJUS mentors for further doubts on the topic.
Put your understanding of this concept to test by answering a few MCQs. Click Start Quiz to begin!
Select the correct answer and click on the Finish buttonCheck your score and answers at the end of the quiz
Recommended Reading: What Is The Molecular Geometry Of Ccl4
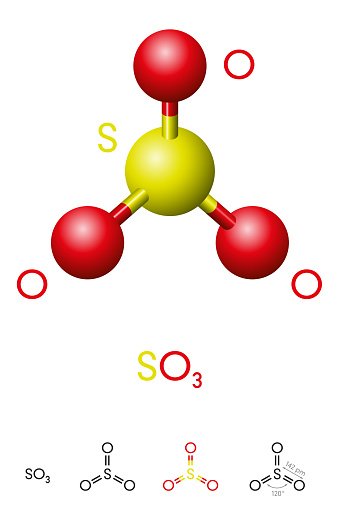
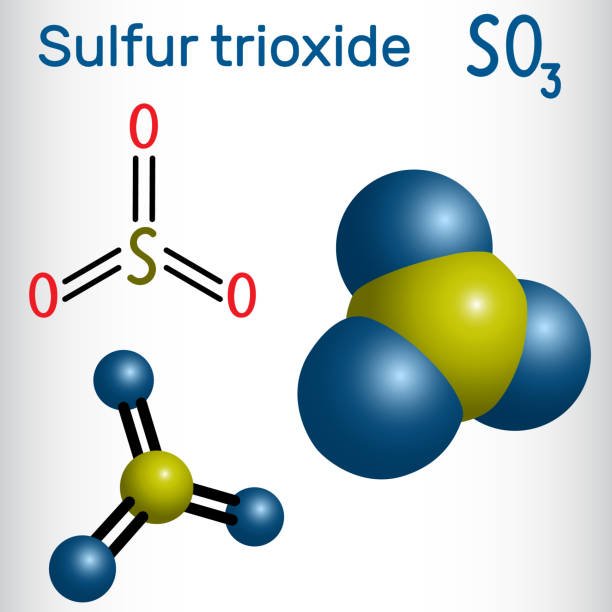
So3 Lewis Structure Molecular Geometry And Hybridization
Sulfur trioxide is a compound with the chemical formula SO3. This compound is of great importance and studied widely as it reacts with the water present in the air to produce sulfuric acid.
When this sulfuric acid, in the gaseous state, mixes with the rain and falls on the Earth, it is called acid rain. The sulfur trioxide is usually colorless and oily but is highly corrosive in nature.
Also, it is called battery acid and is used to manufacture acids, fertilizers, lead-acid batteries, in the pickling of metal, purification of petroleum, and others.
The lewis structure is also called an electron dot structure which determines the number of valence electrons present in an atom.
Moreover, they also describe how these valence electrons are participating in the bond formation to form a molecule.
Also, the Lewis structure helps with figuring out the number and nature of the bond shown with the help of lines.
This structure is an integral component for anyone who is beginning to study atomic chemistry.
To understand these diagrams, it is essential to know how the number of valence electrons has been calculated. The atomic number of the sulfur is 16 which makes its electronic configuration 1s2 2s2 2p6 3s2 3p4.
As the p shell needs to accommodate 6 electrons, there is a need for two more electrons to complete the 3p shell. On the other hand, the atomic number of oxygen is eight that make its electronic configuration 1s2 2s2 2p4.
So3 Molecular Geometry Lewis Structure And Polarity Explained
SO3 stands for Sulfur Trioxide. This is one of the most pollutant chemical compounds in the gaseous form. It is also a primary agent in the acid rain. The main use of this component is to make sulfuric acid for industrial purposes.
SO3 which is also spelled as Sulphur Trioxide sometimes, is a trigonal planar molecule that is non-flammable. In this article, I will provide you some information regarding SO3 molecular geometry with the explanations of Lewis structure, polarity, and hybridization.
Contents
Also Check: What Is The Molecular Geometry Of Ccl4
What Gets Stored In A Cookie
This site stores nothing other than an automatically generated session ID in the cookie; no other information is captured.
In general, only the information that you provide, or the choices you make while visiting a web site, can be stored in a cookie. For example, the site cannot determine your email name unless you choose to type it. Allowing a website to create a cookie does not give that or any other site access to the rest of your computer, and only the site that created the cookie can read it.
Uses Of Sulfur Trioxide So3
- Used as a bleaching agent to remove residual hydrogen peroxide, or as a digesting agent to separate pulp from lignin.
- Used as a catalyst in the oxidation reaction of sulfur dioxide to sulfur trioxide.
- It is a strong inorganic acid mists containing sulfuric acid used in industry or in production of commercial products.
- Used as an essential reagent in sulfonation reaction.
- Used in the manufacture of photoelectric cells and solar energy devices.
Put your understanding of this concept to test by answering a few MCQs. Click Start Quiz to begin!
Select the correct answer and click on the Finish buttonCheck your score and answers at the end of the quiz
You May Like: Holt Geometry Lesson 4.5 Practice B Answers
Use And Labeling Regulations
In 1986, the U.S. Food and Drug Administration banned the use of sulfites as preservatives on foods intended to be eaten fresh . This has contributed to the increased use of erythorbic acid and its salts as preservatives.
Generally, U.S. labeling regulations do not require products to indicate the presence of sulfites in foods unless it is added specifically as a preservative; still, many companies voluntarily label sulfite-containing foods. Sulfites used in food processing are required to be listed if they are not incidental additives ), and if there are more than 10 ppm in the finished product )
Products likely to contain sulfites at less than 10 ppm do not require ingredients labels, and the presence of sulfites usually is undisclosed.
In Australia and New Zealand, sulfites must be declared in the statement of ingredients when present in packaged foods in concentrations of 10;mg/kg or more as an ingredient; or as an ingredient of a compound ingredient; or as a food additive or component of a food additive; or as a processing aid or component of a processing aid.
Sulfites that can be added to foods in Canada are potassium bisulfite, potassium metabisulfite, sodium bisulfite, sodium dithionite, sodium metabisulfite, sodium sulfite, sulfur dioxide and sulfurous acid. These can also be declared using the common names sulfites, sulfates, sulfiting agents.
In the European Union, “EU law requires food labels to indicate contains sulfites without specifying the amount”.
Structure Of Solid So3
The nature of solid SO3 is a surprisingly complex area because of structural changes caused by traces of water. Upon condensation of the gas, absolutely pure SO3 condenses into a trimer, which is often called -SO3. This molecular form is a colorless solid with a melting point of 16.8 °C. It adopts a cyclic structure described as 3.
If SO3 is condensed above 27 °C, then -“SO3” forms, which has a melting point of 62.3°C. -SO3 is fibrous in appearance, like asbestos . Structurally, it is the polymer n. Each end of the polymer is terminated with OH groups . -SO3, like the alpha form, is fibrous but of different molecular weight, consisting of an hydroxyl-capped polymer, but melts at 32.5 °C. Both the gamma and the beta forms are metastable, eventually converting to the stable alpha form if left standing for sufficient time. This conversion is caused by traces of water.
Relative vapor pressures of solid SO3 are alpha< beta< gamma at identical temperatures, indicative of their relative molecular weights. Liquid sulfur trioxide has vapor pressure consistent with the gamma form. Thus heating a crystal of -SO3 to its melting point results in a sudden increase in vapor pressure, which can be forceful enough to shatter a glass vessel in which it is heated. This effect is known as the “alpha explosion.”
Also Check: Paris Jackson Biological Father
What Is The Name Of The Covalent Compound So3
Sulfur trioxide is the chemical compound with the formula SO3.
What is SO3 compound name?,
| Code | |
| Program Version Name ACDLabs 10.04 sulfite OpenEye OEToolkits 1.5.0 sulfite | |
| Formula | |
| Formal charge | -2 |
Furthermore, What is the name of the covalent compound SO3 SO 3?, This means the name of SO3 is sulfur trioxide.
Finally,; What is the name of the following covalent molecule SO3?,
| A |
Lewis Structure Of So3
The sulfur trioxide is a tetra atomic chemical molecule where both the sulfur and three oxygen molecules bond with an equal number of valence electrons.
The diagram is drawn showing dots of valence electrons around the symbol of both sulfur and oxygen atoms with lines predicting bond formation.
The Lewis structure of sulfur trioxide molecule is drawn by:
First, look for the total number of valence electrons in a single sulfur trioxide molecule, which is twenty-four.
Next, look for how many more valence electrons are needed to complete all the octet in the sulfur trioxide molecule.
It is six for a single sulfur trioxide molecule, where both sulfur and each oxygen atom need two valence electrons to stabilize their atom.
Next, look for the number and type of bonds forming within a single sulfur trioxide molecule. It is three double covalent bonds between sulfur and oxygen atom each.
Lastly, look for the central atom, which will be sulfur in this case. Finally, draw the skeleton as:
Recommended Reading: Elimination Method Steps
Sulfur Trioxide Molecular Geometry
Being an intelligent and well-practiced human being, you must know what is molecular geometry, but let me revise it for the all young students out there. Molecular geometry is the three-dimensional structure of the atoms which helps in the constitution of a molecule. It can determine reactivity, polarity, color, attraction, biological activity, etc.
SO3 includes two components mainly Sulfur and Oxygen. There are one sulfur atom and three oxygen atoms which are spread out as far away as they can! Atoms of oxygen are surrounded by electrons. These electrons are negative and repel each other.
You can also remember it by AXN. Where;
- A stands for Sulfur, which is central atom
- X stands for No. of atoms bonded with central sulfur
- N stands for any nonbonding electron pairs
In this formula of SO3, we dont have any non-bonding electron, and that is why we dont bother about N. Moreover, as there are three oxygen, it will be X3.
That means we have AX3 for the SO3 molecule. The bond angle of SO3 is 120 degrees.
Which Has Shorter Bonds So3 Or So3 2
Explanation: SO3 molecule contains 6sigma bonds and bonds to the three O atoms. The average bond order of an S-O bond becomes 6/3=2. In SO3-2 ion, there is a total of four sigma bonds and bonds to the three O atoms. Since bond length decreases as bond order increases, SO3 have a shorter bond length.
Also Check: Eoc Fsa Warm Ups Algebra 1 Answers
Molecular Structure And Bonding
The molecule SO3 is trigonal planar. As predicted by VSEPR theory, its structure belongs to the D3hpoint group. The sulfur atom has an oxidation state of +6 and a formal charge of 0. The S-O bonding is delocalized with all three S-O bond lengths equal at 1.42 Å. The electrical dipole moment of gaseous sulfur trioxide is zero.
Sulfur trioxide undergoes many reactions.
- Hydration and hydrofluorination
SO3 is the anhydride of H2SO4. Thus, it is susceptible to hydration:
- SO3 + H2O H2SO4 ” rel=”nofollow”>mol1) Gaseous sulfur trioxide fumes profusely even in a relatively dry atmosphere owing to formation of a sulfuric acid mist.
Akin to the behavior of H2O, hydrogen fluoride adds to give fluorosulfuric acid:
- SO3 + HF FSO3H
SO3 reacts with dinitrogen pentoxide to give the nitronium salt of pyrosulfate:
- 2 SO3 + N2O5 2S2O7
- Oxidant
Sulfur trioxide is an oxidant. It oxidizes sulfur dichloride to thionyl chloride.
- SO3 + SCl2 SOCl2 + SO2
- Lewis acid
SO3 is a strong Lewis acid readily forming adducts with Lewis bases. With pyridine, it give the sulfur trioxide pyridine complex. Related adducts form from dioxane and trimethylamine.
- Sulfonating agent
Sulfur trioxide is a potent sulfonating agent, i.e. it adds SO3 groups to substrates. Often the substrates are organic. For activated substrates, Lewis base adducts of sulfur trioxide are effective sulfonating agents.
The direct oxidation of sulfur dioxide to sulfur trioxide in air proceeds very slowly:
- SO2 +
Which Catalyst Is Used In Contact Process
The contact process is the current method of producing sulfuric acid in the high concentrations needed for industrial processes. Platinum used to be the catalyst for this reaction. However, as it is susceptible to reacting with arsenic impurities in the sulfur feedstock, vanadium oxide is now preferred.
You May Like: What Does Abiotic Mean In Biology
Main Difference So2 Vs So3
SO2 and SO3 are inorganic chemical compounds formed by the combination of sulfur atoms and oxygen atoms. SO2 stands for sulfur dioxide, and SO3 stands for sulfur trioxide. These are gaseous compounds. They have different chemical and physical properties. These compounds are called oxides of sulfur since they are formed from the reaction between sulfur and O2 molecules. The main difference between SO2 and SO3 is that SO2 has two oxygen atoms bonded to a sulfur atom whereas SO3 has three oxygen atoms bonded to a sulfur atom.