Properties Of Homogeneous Mixtures
Homogeneous mixtures have several identifying properties:
Saltwater is an example of a homogeneous mixture that can be easily separated by evaporation.
Many countries use evaporation to capture pure drinking water from seawater, leaving behind salt that can be sold for profit.
Sugar water is another example of a homogeneous mixture that can be separated by evaporation.
How To Pronounce Homogenous And Homogeneous
These words have different pronunciations, and pronouncing each word correctly is key to using the correct word in your writing.
How to pronounce homogenous: The pronunciation of homogenous has four syllables.
How to pronounce homogeneous: The pronunciation of homogeneous has five syllables. Homogeneous, along with its antonym, heterogeneous, both contain more syllables than homogenous.
- Homogenous = four syllables.
- Heterogeneous = six syllables.
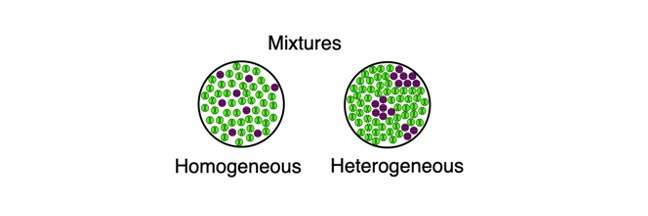
What Are Homogeneous And Heterogeneous Mixtures
Homogeneous mixtures are the mixtures that have a uniform composition of substances throughout the mass. In other words, the particles in the homogeneous mixtures are uniformly distributed throughout the composition. Due to the uniformity of the mixture, it is often confused with pure solutions. But, unlike pure solutions, homogeneous mixtures can easily be separated by the precipitation method as the particles do not react with each other.
On the other hand, a heterogeneous mixture can be defined as a mixture that has an uneven composition throughout the mass. Alternatively, it is stated as the mixture in which the particles are not uniformly distributed throughout the composition and can easily be identified with the naked eye or under a microscope. Heterogeneous has two phases. For example, when oil and water are mixed together, due to the low density of oil, it starts floating above the water surface. Thus, this mixture comprises two phases, one of water and the other of oil.
The Major Differences Between Homogeneous and Heterogeneous Mixture are:-
Recommended Reading: What Is Apa In Psychology
What Is Homogeneous Environment In Ecology
In ecology, species homogeneity is a lack of biodiversity. Species richness is the fundamental unit in which to assess the homogeneity of an environment. Therefore, any reduction in species richness, especially endemic species, could be argued as advocating the production of a homogenous environment.
Distribution Of Containers In The Matrices Of Novel Protective Coatings

Distribution of containers in the matrix of the cured coating is closely related to their dispersibility in the initial coating formulation. This characteristic, in turn, depends on the interplay between the interaction of containers with themselves and with solvent components of the coating formulation . If the work of adhesion exceeds the work of cohesion the wetting of containers by the coating formulation medium is energetically disadvantageous and they tend to form aggregates. In the best case of lower adhesion work, the good wetting of containers is achieved and they may be dispersed well in the coating formulation. Other important preconditions for stability of containers dispersion are electrostatic or steric barriers around containers preventing their aggregation. On the other hand, such factors as high density difference between material of containers and the formulation medium as well as presence of other particles , such as particles of the color pigment, can lead to the destabilization of the entire coating formulation even prior application and then to the very inhomogeneous distribution of containers in the matrix of the cured coating.
At the same time, the polar nature of solvent-free powder coatings allowed the homogeneous distribution of not only containers with PUa shells, but also of much more polar halloysites-based containers .
Eliana B. Bergter, Maria Helena S. Villas Boas, in, 1995
Recommended Reading: What Does Endpoint Mean In Geometry
What Does Homogenous Mean
Homogenous most generally means consisting of parts or elements that are all the same. Something that is homogenous is uniform in nature or character throughout.
Homogenous can also be used to describe multiple things that are all essentially alike or of the same kind. When used generally, homogenous is often associated with things that are considered biased, boring, or bland due to lacking variety or diversity.
In the context of chemistry, homogenous is used to describe a mixture that is uniform in structure or composition. The verb homogenize means to make something homogenous. To homogenize milk is to blend it in a way that results in the milk fat being evenly distributed throughout it. The state of being homogenous is homogeneity.
The general sense of homogenous can be used interchangeably with the word homogeneous . When used in this general way, homogenous is more commonly used than homogeneous.
Homogenous is also used in the context of biology to mean corresponding in structure due to either a common origin or a common environment.
The opposite of most senses of homogenous is heterogeneous, which most generally means consisting of different, distinguishable parts or elements.
Example: This suburb is so homogenouseven the houses all look the same.
Differences Between Homogeneous And Heterogeneous Mixture
|
Homogeneous Mixtures |
|
|
Particles are uniformly distributed throughout the mass |
Particles are non-uniform throughout the mass |
|
Homogeneous mixtures have the same composition throughout the mixture |
Heterogeneous mixtures have different compositions throughout the mixture. |
|
The particles cannot be separated physically |
The particles can be separated physically |
|
It has a single-phase throughout the mixture |
It has two or more phases throughout the mixture |
|
Example: sugar water mixture, salt water mixture |
Example: smog particles in the air, oil-water mixture |
Read Also: What Is Physical Geography For Kids
When To Use Homogeneous
What does homogeneous mean? Homogeneous is an adjective. It is not a synonym for homogenous, despite its spelling similarities to that word.
Homogeneous means having similar or uniform characteristics. A community where most members share similar characteristics, e.g., a biker gang composed of low-income males in their 20s and 30s who all have similar tattoos, could be said to be homogeneous. A box of crayons that only contains blue crayons would also be homogeneous.
Here are a few more examples:
- Cake batter starts as a collection of disparate ingredients, but soon becomes a gooey, homogeneous mixture.
- Sales figures for existing branches vs new branches are remarkably homogeneous there are no statistically significant differences between branches during the most recent quarter.
- The net result is parties that are much more internally homogeneous than was the case a generation ago. The Washington Post
Homogeneous has appeared in English since the 17th century. It is somewhat unique in that its etymology can be traced through both Latin and Greek. It should be pronounced with five distinct syllables, hoh-muh-jeen-ee-us.
The associated noun for homogeneous is homogeneity, and its verb is homogenize. You will likely have seen milk labels advertising the product as homogenized. Homogeneous also has an antonym, heterogeneous, that means having dissimilar characteristics.
Homogeneous: Definition And Examples
- Ph.D., Biomedical Sciences, University of Tennessee at Knoxville
- B.A., Physics and Mathematics, Hastings College
“Homogeneous” refers to a substance that is consistent or uniform throughout its volume. A sample taken from any part of a homogeneous substance will have the same characteristics as a sample taken from another area.
Recommended Reading: How To Study For Chemistry In College
Examples Of Homogeneity In A Sentence
homogeneity The New Republichomogeneity Los Angeles Timeshomogeneity Washington Posthomogeneity Robb ReporthomogeneityThe New YorkerhomogeneityNew York Timeshomogeneity The New YorkerhomogeneityThe Christian Science Monitor
These example sentences are selected automatically from various online news sources to reflect current usage of the word ‘homogeneity.’ Views expressed in the examples do not represent the opinion of Merriam-Webster or its editors. Send us feedback.
Introduction To Homogeneous Reaction
Homogeneous chemistry is a broader term that is specifically used in the context of Chemistry involved with homogeneous mixtures or homogeneous solutions. Homogeneous chemistry basically refers to the homogeneous reactions that take place in various conditions. So in order to understand the chemistry of homogeneous reactions, it is important to have a clear knowledge of homogeneous mixtures and solutions.
A homogeneous mixture can be defined as a mixture that has a uniform composition throughout its mass. For example sugar in water or salt in water. That is, when sugar or salt is mixed in water they dissolve completely in the solvent which is water in this case. Many a time, a homogeneous mixture is confused with pure substance as they are both uniform in nature.
The major difference between a homogeneous mixture and a pure substance is that the composition of the pure substance is uniform in all the samples of that particular substance. But, although the composition of a homogeneous mixture is uniform throughout a particular sample, the percentage of the composition will vary from sample to sample.
Though all uniform compositions of various samples are broadly termed as homogeneous mixtures, they can be solid to liquid, solid to solid, liquid to liquid or gas to gas. But homogeneous solution is particularly used to denote solid to liquid mixture or liquid to liquid mixture.
Don’t Miss: What Is Alt Sgpt In Blood Chemistry
What Is Heterogeneity Of Ecosystem
- Heterogeneity can be conceptualized in different ways for different purposes, resulting in multiple definitions and operationalizations. Multiple conceptualizations, definitions, and operationalizations of heterogeneity allow for the investigation of different aspects of ecological patterns on different ecological processes.
Related
What Is Heterogeneity In Ecosystem
Environmental heterogeneity is regarded as one of the most important factors governing species richness gradients. An increase in available niche space, provision of refuges and opportunities for isolation and divergent adaptation are thought to enhance species coexistence, persistence and diversification.
Don’t Miss: Texas Algebra 1 Textbook Mcgraw Hill
What Is Limiting Factor In Ecology
A limiting factor is anything that constrains a population’s size and slows or stops it from growing. … Limiting factors are usually expressed as a lack of a particular resource. For example, if there are not enough prey animals in a forest to feed a large population of predators, then food becomes a limiting factor.
Related
What Is Homogeneous Equilibrium And Heterogeneous Equilibrium
A homogeneous equilibrium is a state in which all the reactants and products are present in one single solution. A homogeneous equilibrium can be established only when the reactions taking place between various solutes in the liquid solutions are in the same homogeneous equilibrium. The chemical species involved in the reaction can be molecules, ions or mixtures. Example,
C2H2 + 2Br2 C2H2B4
Therefore, equilibrium constant will be, K = / 2
A heterogeneous equilibrium is a state in which the reactants and the products of a reaction are in two or more phases. These phases can be in a combination of solid, liquid, gases or solutions.
Recommended Reading: What Does Vo Mean In Physics
What Is A Homogeneous Environment Biology
In ecology, species homogeneity is a lack of biodiversity. Species richness is the fundamental unit in which to assess the homogeneity of an environment. Therefore, any reduction in species richness, especially endemic species, could be argued as advocating the production of a homogenous environment.
Examples Of Homogeneous In A Sentence
Focus on GeographyNew York Times Book ReviewThe Civilization of the Middle AgeshomogeneoushomogeneousForbeshomogeneousNew York Timeshomogeneous The Weekhomogeneous CNNhomogeneous New York TimeshomogeneousThe Hollywood Reporterhomogeneous New York Timeshomogeneous CNN
These example sentences are selected automatically from various online news sources to reflect current usage of the word ‘homogeneous.’ Views expressed in the examples do not represent the opinion of Merriam-Webster or its editors. Send us feedback.
Don’t Miss: Crash Course Geometry John Green
Homogenous Vs Homogeneous Whats The Difference
Home » Homogenous vs. Homogeneous Whats the Difference?
If you are describing a society that is made up of very similar people, would you describe this society as homogenous or homogeneous?
Despite their spelling similarities, these words are not synonyms. One is an outdated term from biology, while the other is an adjective that refers to sameness. They are confusing enough that even professional journalists often misuse them.
You, however, are a discerning writer, and you understand that proper word choice can preserve your credibility. This article will explain which of these words is correct in your writing.
Solid Homogeneous Mixture Examples
Homogeneous mixtures are also known as solutions. When you think of a solution, you probably think of a liquid. However, many solids are also considered homogenous mixtures. There is a wide variety of solid homogeneous mixtures, from naturally occurring materials like stone to synthetic plastics.
- Bitumen – the solid form of petroleum and source of gasoline, diesel and other fossil fuels bitumen is a homogeneous mixture of complex hydrocarbon chemicals
- Cement – a solid homogeneous mixture of calcium compounds mixed with sand, gravel and water, it becomes the heterogeneous mixture concrete, one of the most important building materials in the world.
- Bronze – a mixture of copper and tin bronze is a type of alloy, which is a metal created by two or more metallic elements
- Steel – an alloy made from iron and carbon both steel and stainless steels, which includes chromium, are homogeneous mixtures
- Brass – a metal made with copper and zinc brass is also an alloy
- Thermoplastics – a type of manufactured plastic that includes polyethylene and polyvinyl chloride thermoplastics include polymers that heat and cool uniformly into solid plastic substances
You may be wondering if certain types of stone, such as granite, are homogeneous. Rocks are made out of different minerals, crystals and substances, making them heterogeneous. However, the minerals that compose rocks are often homogeneous themselves.
You May Like: What Do You Call Your Friends In Math Class
What Is The Difference Between Functional And Structural Heterogeneity
- Moreover, from an ecological perspective it is necessary to differentiate between functional and structural heterogeneity. Functional heterogeneity is defined with respect to particular ecological processes. In contrast, structural heterogeneity takes an arbitrary, or observer-oriented, perspective on the assessment of heterogeneity.
What Are Heterogeneous Mixtures

- It is a mixture which has physically distinct parts
- Such mixtures have non uniform composition throughout the mixture
- We are able to recognize individual particles by looking at a mixture
Mixture of Oil and Water is a heterogeneous Mixture
We are able to see oil and water clearly separately in the mixture
Mixture of Salt and Iron filings is a heterogeneous mixture
The particles of salt and Iron filings can be seen and distinguished easily
Read Also: What Does Macro Mean In Biology
Choice Of Materials Methods And Laboratories
Homogeneity and stability must be assured for the study to have any use at all. Usually, one laboratory has the responsibility for performing tests. Subsamples will be stored at different temperatures ranging from, say 20 °C to + 50 °C and then analyzed once a month.
Use of a single method by a small number of laboratories runs the risk of introducing a method bias into the result. It is recommended that the laboratories are chosen for their high standard of analytical capability, and their ability to apply different methods, where this is appropriate. If necessary the laboratories will also be asked to employ different pretreatment methods. A large number of laboratories should be chosen, with 20 in the initial phase of the certification.
R. Alagirusamy, in, 2010
Trick To Remember The Difference
Unless you are a 19th century biologist, you should avoid using homogenous. Today, scientists would be more likely to use homologous in place of this archaic term.
For all situations where you need to describe something as having similar or uniform characteristics, choose homogeneous.
Homogeneous vs. Homogenous Check: Since homogeneous has an extra E, like the word characteristics, you can remember that homogeneous describes similar characteristics.
Read Also: What Is The Law Of Conservation Of Momentum In Physics
When To Use Homogenous
What does homogenous mean? Homogenous is an outdated biological term that refers to organs or body tissues with genetic similarities.
Today, most scientific writers would use the more recent term homologous instead.
Still, if you are reading older scientific texts, you might run across homogenous from time to time.
Here is a possible example:
- The host body rejected even the homogenous tissue implants.
The usual mistake with these words is to use the word homogenous when the writer really means homogeneous. Since homogenous is declining in use, even in the scientific community, this should be an easy error to avoid.
Why Is Spatial Heterogeneity Important
Spatial heterogeneity is important for point processes when the average rate must be determined over an area that is spatially heterogeneous or understanding or predicting the spatial pattern of process rates is an objective, for example, to identify areas of high or low rates, or to quantify the spatial …
Related
Read Also: Which University Housed The World’s First Psychology Laboratory
Examples Of Homogeneous Mixtures: Solid Liquid And Gas
A chemical mixture combines two substances that maintain their own properties when combined. Heterogeneous mixtures are made up of a non-uniform composition, while homogeneous mixtures are made up of a uniform composition. For example, water and sand is a heterogeneous mixture you can easily separate the sand from the water. But orange juice is homogenous it would be difficult, if not impossible, to separate the orange particles from the water. Keep reading for more examples of solid, liquid and gaseous homogenous mixtures that you see every day.