What Is A Dl Pair In Stereochemistry
Is it two isomers with at least one stereogenic center each but the corresponding center on the other isomer will rotate the plane of polarized light in the opposite direction. Is this applicable to meso compounds which aren’t chiral overall but have stereogenic centers?
d-l- Nomenclature as well as , refer to a physical property of the molecule, its optical rotation. Optical rotation is a result of chirality, a topological property of the molecule. So d-, l- is something that you can generally measure with a polarimeter. If you have a 1:1 mixture of the 2 enantiomers then you will see zero rotation and this mixture is called racemic and named with the dl- prefix. A meso compound also has zero optical rotation since it is achiral but as it is not a mixture the dl- notation doesn’t make sense but you might see it used sometimes.
The problem now is if you are given a pair of enantiomeric molecules that are mirror images how do you name them so you can tell them apart every time? This is why the D-, L- and R-, S- notations were introduced. D- L- is the older and more naive approach that was later improved by the R-, S- notation. Unfortunately as chemistry is a science with long history that is also used by other non chemistry-trained professionals, several times older naming conventions have survived. So you have to know all of them!
What Is The Meaning Of Dl Abbreviation In Chemistry
What is DL definition ?
What does DL mean in Chemistry?
DL mean that “Detection Limit” for Chemistry.
What is DL acronym ?
What is shorthand of Lethal Dose ?
The shorthand of “Lethal Dose” is DL.
What is the definition of DL acronym in Chemistry?
Definitions of DL shorthand is “Deciliter”.
What is the full form of DL abbreviation?
Full form of DL abbreviation is “Detection Limit”.
What is the full meaning of DL in Chemistry?
Full meaning of DL is “Lethal Dose”.
What is the explanation for DL in Chemistry?
Explanation for DL is “Deciliter”.
What is the meaning of DL Abbreviation in Astrology ?
The site does not only include the meanings of the DL abbreviation in Chemistry. Yes, we know your main purpose is explanation of DL abbreviation in Chemistry. However, we thought that besides the meaning of the DL definitions in Chemistry, you can consider astrological information of DL acronym in Astrology. Therefore, the astrological explanation of each word in each DL abbreviation is also included.
DL Abbreviation in Astrology
What Is The Difference Between D And L Configuration And + And
What is the difference between D and L configuration, and + and ?
My textbook says they are two different things. It also says that the correct way to name glucose is D-glucose.
Could someone please explain what D, L and +, represent, and why they are different?
The D-L system corresponds to the configuration of the molecule: spatial arrangement of its atoms around the chirality center.
While and notation corresponds to the optical activity of the substance, whether it rotates the plane of polarized light clockwise or counterclockwise .
D-L system tells us about the relative configuration of the molecule, compared to the enantiomers of glyceraldehyde as the standard compound. Compounds with the same relative configuration as -glyceraldehyde are assigned the D prefix, and those with the relative configuration of -glyceraldehyde are given the L prefix.
It’s kind of another way to tell the configuration of molecules beside the CahnIngoldPrelog convention , with little difference.
In short, the D-L system doesn’t have direct connection to / notation. It only relates the stereochemistry of the compound with that of glyceraldehyde, but says nothing about its optical activity. We may have compound with same relative configuration as -glyceraldehyde , yet it rotates the polarized light counterclockwise , such as D–ribose.
And also, don’t confuse the D-L system with d- and l- naming. d- and l- is the exact same with and notation.
Additional explanation
Reference
You May Like: Answers To Odysseyware Algebra 1
Background Of D/l Notation
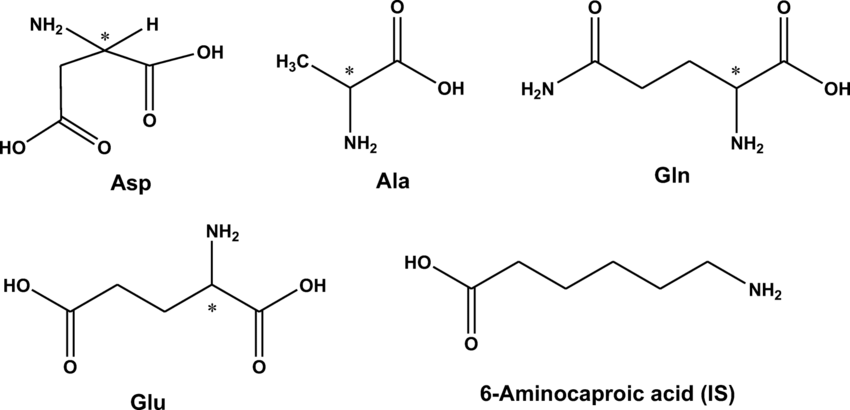
The d/l system names molecules by relating them to the molecule glyceraldehyde. Glyceraldehyde is chiral, and its two isomers are labeled d and l . Certain chemical manipulations can be performed on glyceraldehyde without affecting its configuration, and its historical use for this purpose has resulted in its use for nomenclature. In this system, compounds are named by analogy to glyceraldehyde, which, in general, produces unambiguous designations, but is easiest to see in the small biomolecules similar to glyceraldehyde. One example is the chiral amino acid alanine, which has two optical isomers, and they are labeled according to which isomer of glyceraldehyde they come from. On the other hand, glycine, the amino acid derived from glyceraldehyde, has no optical activity, as it is not chiral .
The d/l labeling is unrelated to / it does not indicate which enantiomer is dextrorotatory and which is levorotatory. Rather, it says that the compound’s stereochemistry is related to that of the dextrorotatory or levorotatory enantiomer of glyceraldehydethe dextrorotatory isomer of glyceraldehyde is, in fact, the d- isomer. Nine of the nineteen l-amino acids commonly found in proteins are dextrorotatory , and d-fructose is also referred to as levulose because it is levorotatory.
Priority Rules Of Rs System
First Rule
- Atoms with the highest atomic number are given priority.
- Atoms with the lowest atomic number are given the lowest priority.
Second Rule
- A clockwise rotation yields R as the configuration of the chiral center, from the Latin rectus, which means right.
- The chiral center of a circle is shaped like an S, based on the Latin sinister, which means “left”.
Also Check: Solving Exponential Equations Algebra 2
Dl System Of Nomenclature Of Optimal Isomers
- In Fischer projection, carbonyl groups will be positioned on top, and CH2OH will be positioned on the bottom.
- For carbon 2, the OH or NH2 is used as the D descriptor when it is on the right side, while the L descriptor is used when it is on the left side. Thus, it is possible to determine the stereochemistry of all other stereocenters in a molecule by the stereochemistry of its second carbon from the bottom.
- Dextrorotatory and levorotatory enantiomers are not specified by the d/l nomenclature. A similarity can be found between its stereochemistry and that of the levo or dextro enantiomer of glyceraldehyde. Levorotatory substances, such as d-fructose, are examples. Thus, all natural amino acids have the L configuration, whereas all natural carbohydrates have the D configuration. In this sense, glucose has a D-configuration, while ribose has an L-configuration.
Determining D/l Notation In Carbohydrates
A Fischer projection is used to differentiate between L- and D- carbohydrates. On a Fischer projection of a monosaccharide, the penultimate carbon of D sugars are depicted with hydrogen on the left and hydroxyl on the right. L sugars will be shown with the hydrogen on the right and the hydroxyl on the left. In a standard Haworth projection of the cyclohexane structures of hexoses, D-carbohydrates have the terminal carbon pointing up.
Also Check: What Is Regression To The Mean In Psychology
Optical Activity And Dl Nomenclature
The two isomers – and – of a compound exhibit optical activity . However, this nomenclature does not indicate whether the compound is dextrorotatory or left- handed , that is, if it deviates the plane of polarized light to the right or to the left, although it may coincide in some cases. For example, D-glyceraldehyde is dextrorotatory , that is, the two forms of glyceraldehyde are – – glyceraldehyde and – – glyceraldehyde. But this is not always the case. – – lactic acid is left-handed , while – – lactic acid is right-handed .
Dl Meaning In Chemistry
Please also find DL meaning for Chemistry in other sources.
- And finally again and again search .
You May Like: What Is Substitution In Biology
What Is D And L Convention
The D,L convention, not to be confused with the d and l descriptors used to designate the direction of specific rotation of chiral compounds, is a convention used to distinguish between enantiomers of chiral monosaccharides and chiral alpha-amino acids, based on the molecule drawn as a Fischer projection in a specific
You Are Reading A Preview
Activate your 30 day free trial to continue reading.
This a very interesting topic from Chemistry, mainly STEREO-CHEMISTRY….
This a very interesting topic from Chemistry, mainly STEREO-CHEMISTRY….
Also Check: What Is Latent Learning In Psychology
What Does Dl Stand For Chemistry
We compiled queries of the DL abbreviation in Chemistry in search engines. The most frequently asked DL acronym questions for Chemistry were selected and included on the site.
We thought you asked a similar DL question to the search engine to find the meaning of the DL full form in Chemistry, and we are sure that the following Chemistry DL query list will catch your attention.
What Is Difference Between D And L
Summary L vs D Isomers
The main difference between L and D isomers is in the position of OH group in the penultimate carbon atom. In D isomer, the OH- group of the penultimate carbon is positioned on the right side whereas, in L isomer, the OH- group of the penultimate carbon is positioned on the left side.
Also Check: Infinite Algebra 1 One Step Equations
In Electronics And Computing
- < dl> < /dl> , an HTML element used for a definition list
- Deep learning, a branch of algorithm-based machine learning
- Description logics, a family of knowledge representation languages
- Delete Line , an ANSI X3.64 escape sequence
- Digital library, a library in which collections are stored in digital formats
- Diode logic, a logic family using diodes
- DVD-R DL, a DVD Dual Layer engineering method
- DL register, the low byte of an X86 16-bit DX register
- Dynamic loading, a mechanism for a computer program to load a library
- Data link, a computer connection for transmitting data
- Distribution list, a function of e-mail clients
- Downlink, the link from a satellite to a ground station
- , a transfer of electronic data
Limitations Of The Sugar Convention
- There are multiple chirality centers in sugar molecules. The configuration of the chirality center with the highest number is shown, but that of the other centers is not shown .
- In the conversion of compounds from one series to another, another complication occurs. D-configuration is assigned to saccharic acid, which is obtained from D-glucose through oxidation. The carbon with the highest number of chiral configurations is D. The highest chiral configuration then is L, which is formula 25. However, there is a serious problem with this approach, in that the same molecule can possess both D- and L- configurations.
- It may be necessary to designate -tartaric acid and -threonine as D or L depending on whether the reference compound is glyceraldehyde or hydroxyl or amino acid . Additionally, this system is limited to sugars, amino acids, and hydroxy acids and is therefore of limited use.
- Hydroxy acid or amino acid convention
Read Also: What Is Sample Space In Math Terms
Normal Values For Lab Tests
Lab tests, along with a health history and physical exam, are used by physicians to diagnose and manage health conditions. When your body is healthy, it functions normally in a state of homeostasis or equilibrium. When your body is in homeostasis, the values for fluids, chemicals, electrolytes, and secretions are typically within an acceptable or normal range. When you have a pathologic condition , these ranges can change as a result of an imbalance in your body. The disease can cause the imbalance, and vice versa, the imbalance may cause the disease. Lab tests can help identify these imbalances which will aid the medical professional in understanding and treating the underlying problem.
Furthermore, if you are on medications, lab tests and lab profiles can also be used to determine the exact amount of medicine that you should be taking to appropriately treat your condition. The normal range of values for lab tests are established and acceptable values within which the test results for a healthy person are expected to fall.
| 1.2 1.3 mg/dL or less |
| Blood group |
| male: 17 148 U/L, female: 10 70 U/L |
| Creatinine, serum |
| male: 0 15 mm/hr, female: 0 20 mm/hr |
| Sodium, serum |
| male: 3.9 9 mg/dL, female: 2.2 7.7 mg/dL |
| Urinalysis, routine |
What Does Dl
I came across the compound dl-stilbene dibromide, and I am confused by exactly what this means. I thought it might have meant a racemic mixture but there is only one chemical structure given.
That picture is showing relative stereochemistry. Essentially, it is just saying that, in the shown confirmation, the hydrogens will be on one side and the bromines will be on the other, but half of the mixture will have the bromines coming out and the other half will have the bromines going back.
If the S or R labels had been shown in the molecule the picture would have been showing absolute stereochemistry. By saying dl-stilbene dibromide, they are saying that it is a racemic mixture of the compound shown and its enantiomer, but not any diastereomers.
Here’s a snippet with surroundings from the book , for the context:
Should we accept the name stilbene dibromide ), there are some other problems.
49 is defined in the text as a diastereomer, but the reaction scheme it’s named dl-Stilbene dibromide, denoting a racemic mixture , therefore should apply to entire 49 structure including the text.
It’s true that sometimes the constant-width bold/hashed bond is used to denote relative stereochemistry, together and in contrast with wedge bold/hashed bonds for absolute stereochemistry. This mixed convention is not supported by IUPAC, even it’s described as obscure in Graphical representation of stereochemical configuration :
Recommended Reading: Algebra Nation Test Yourself Answers Section 8
What Are L
All amino acids except for glycine are stereoisomers . These are labeled L-amino acids and D-amino acids to distinguish. Most of the amino acids that are manufactured today are L-amino acids.
Amino acids refers to compounds whose molecules contain an amine group and carboxyl group . All amino acids except for glycine are stereoisomers. This means that there are mirror images of their structure. It is just like how we have left hands and right hands. These are labeled L and D to distinguish the mirror images. For some reason, the amino acids that make up the proteins in our bodies are all L-amino acids. Originally, people thought that D-amino acids did not exist in the natural world. However, scientists recently discovered that they do exist and perform a variety of functions. DL-amino acids contain both L- and D-amino acids, and are also called racemic mixtures.
As it is mentioned above, most of the amino acids that are manufactured today are L-amino acids. This includes glutamate, which is used as an umami seasoning. All amino acids referred to here are L-amino acids, unless we state otherwise.