What Are Monoacidic Diacidic And Triacidic Bases
- A monoacidic base is a base that produces one hydroxide ion when one of its molecules undergoes complete ionisation. Examples of such bases include potassium hydroxide and sodium hydroxide.
- A diacidic base is a base that produces two hydroxide ions when one of its molecules undergoes complete ionisation. Examples of such bases include magnesium hydroxide and barium hydroxide.
- A triacidic base is a base that produces three hydroxide ion when one of its molecules undergoes complete ionisation. Common examples of triacidic bases include iron hydroxide and aluminium hydroxide
To learn more about the definition and meaning of bases along with other important concepts related to acids and bases, register with BYJUS and download the mobile application on your smartphone.h BYJUS and download the mobile application on your smartphone.
Put your understanding of this concept to test by answering a few MCQs. Click Start Quiz to begin!
Select the correct answer and click on the Finish buttonCheck your score and answers at the end of the quiz
What Do Chemists Do
Chemists work in a variety of fields, including research and development, quality control, manufacturing, environmental protection, consulting and law. They can work at universities, for the government or in private industry, according to the ACS .
Here are some examples of what chemists do:
Research and development
In academia, chemists performing research aim to further knowledge about a particular topic, and may not necessarily have a specific application in mind. Their results, however, can still be applied to relevant products and applications.
In industry, chemists in research and development use scientific knowledge to develop or improve a specific product or process. For example, food chemists improve the quality, safety, storage and taste of food pharmaceutical chemists develop and analyze the quality of drugs and other medical formulations and agricultural chemists develop fertilizers, insecticides and herbicides necessary for large-scale crop production.
Sometimes, research and development may not involve bettering the product itself, but rather the manufacturing process involved in making that product. Chemical engineers and process engineers devise new ways to make the manufacturing of their products easier and more cost effective, such as increasing the speed and/or yield of a product for a given budget.
Environmental protection
What Are Alkalis How Are They Different From Bases
Alkalis are bases that are soluble in water. Usually, the term alkali is used to denote the ionic salts of alkaline earth metals or alkali metals that exhibit basic qualities. Therefore, it can be understood that alkalis form a subset of all bases. This implies that all alkalis are bases but all bases are not alkalis. It can also be noted that all alkalis adhere to the Arrhenius definition of basic substances .
Some common examples of alkali salts are listed below.
- Calcium hydroxide 2)
- Sodium hydroxide
- Potassium hydroxide
It can also be noted that ammonia is sometimes excluded from the alkali family despite the fact that it facilitates the formation of hydroxide ions when dissolved in water.
Recommended Reading: What Does Algorithm Mean In Math
What Does Chemistry Mean
As an introduction to chemistry, it is the branch of science that studies matter and change. First, chemistry deals with the study of the composition and the properties of matter . Then, chemistry deals with change, or how these substances evolve when submitted to certain conditions, or how one substance changes or reacts while interacting with a different substance. The definition of chemistry cant be made shorter, since it covers basically everything!
Relationship Between Chemistry And Other Branches Of Science

Science can be defined as the systematic study of the natural universe, its structure, and everything it encompasses. Due to the immensity of the natural universe, science has been divided into several disciplines that deal with certain aspects of the universe. The three primary subcategories of science under which these disciplines can be grouped are:
- The Formal Sciences: Involves the study of the language disciplines that concern formal systems. Examples of scientific disciplines that fall under this category include logic and mathematics. Can be thought of as the language of science.
- The Natural Sciences: Involves the study of natural phenomena through experiments and observations. Chemistry, physics, and biology fall under this category of science.
- The Social Sciences: Involves the study of human societies and the relationships between the humans that dwell in these societies. Examples of scientific disciplines that fall under this category include psychology, sociology, and economics.
When the relationships between the major branches of science are considered, chemistry is found to lie close to the centre .
Thus, chemistry can be viewed as a central science whose roots bore into several other subdisciplines of science.
Read Also: What Is Atomic Radius In Chemistry
What Is The Basic Definition Of Chemistry
Chemistry. Chemical science redirects here. For the Royal Society of Chemistry journal, see Chemical Science .
Sometimes the distinction between phases can be continuous instead of having a discrete boundary in this case the matter is considered to be in a supercritical state. When three states meet based on the conditions, it is known as a triple point and since this is invariant, it is a convenient way to define a set of conditions.
Video advice: Matric part 1 Chemistry, Basic Definitions Chemistry Ch 1 9th Class Chemistry
ilmkidunya.com has brought to you Lecture of Sibghat Ullah on \9th Class Chemistry Chapter 1 Fundamentals of Chemistry .
In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both basic and applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant chemistry , the formation of igneous rocks , how atmospheric ozone is formed and how environmental pollutants are degraded , the properties of the soil on the moon , how medications work , and how to collect DNA evidence at a crime scene .
List Some Examples Of Strong Bases
Some examples of strong bases are, Lithium Hydroxide, Sodium Hydroxide, Potassium Hydroxide, Rubidium Hydroxide, Caesium Hydroxide, and Calcium Hydroxide
Bases typically have bitter tastes and soapy textures. The pH values of basic solutions are always above 7. Bases react with acids to form salts. Such a chemical reaction is called an acid-base neutralisation reaction. It can be noted that the pH of an aqueous solution of the salt formed from an acid-base neutralisation reaction is dependent on the strength of the parent acid and the parent base. For example, a strong acid and a weak base will typically yield an acidic salt and a strong base, when reacted with a weak acid, will often yield a basic salt.
You May Like: Pre Algebra With Pizzazz Never Say Die
General Properties Of Basic Substances
- Typically, bases are bitter to the taste.
- Under standard conditions, the pH value of a basic solution is always greater than 7.
- Concentrated solutions of relatively strong bases are known to react with acidic substances in a violent manner. Such substances must, therefore, be stored with caution.
- Powerful bases are also known to have caustic effects on organic matter. Great care must be taken while handling such substances since accidental spillage on the skin can result in severe burns and permanent damage to the tissue.
- All basic substances have similar effects on certain chemical indicators. For example, all bases have the ability to turn red litmus blue. They are also known to impart a yellow colour to methyl orange and pink colour to phenolphthalein.
- In their molten or aqueous states, bases are great conductors of electric currents .
What Is The Future Of Chemistry
Chemistryis the science that studies and manipulates matter. We human beings are gettingpretty good at it, , but we arefar from an ideal position in which we can easily make any molecule or compoundat will in a matter of minutes.
That isprobably the future of chemical synthesis, being able to shape any compound atwill in a matter of minutes, without relying on long term challenging synthesis projects. Furthermore, the possibilities ofsynthetic chemistry are literally endless: there will always be room for makinga chemical even better, or finding a molecule that works even better for agiven task.
Another keyaspect of the chemistry of the future will be reaching true fullsustainability. Chemistry will be one of the main branches of science to solvethe problem of energy.
Also,chemistry, as the central science, will be responsible to helping technologyand interdisciplinary science in general to develop smoothly.
Don’t Miss: What Is Translatory Motion In Physics
Chemistry Is Everywhere: In The Morning
Most people have a morning ritual, a process that they go through every morning to get ready for the day. Chemistry appears in many of these activities.
These are just a few examples of how chemistry impacts your everyday life. And we havent even made it to lunch yet!
View the video The Chemical World by Dr. Jessie A. Key for an introduction to the science of chemistry and how it fits into our everyday lives.
Key Takeaways
- Chemistry is the study of matter and its interactions with other matter and energy.
- Matter is anything that has mass and takes up space.
- Matter can be described in terms of physical properties and chemical properties.
- Physical properties and chemical properties of matter can change.
- Matter is composed of elements and compounds.
- Combinations of different substances are called mixtures.
- Elements can be described as metals, nonmetals, and semimetals.
Exercises
Essential Basic Chemistry Concepts Explained
Learning the basic chemistry concepts, in which an entire chemical education process is based on, can be overwhelming for beginners.
One of the reasons is the vast amount of information that there is out there.
For that reason, I decided to go ahead and explain here 15 important basic chemistry concepts, which hopefully will get you in a better shape for learning chemistry. These are clearly explained in most of the chemistry textbooks in our review.
In case you are starting to learn organic chemistry, we have also published an overview of the most important concepts that you will need, and a further review comparing SN1 and SN2 reactions.
We will start off with an introduction to basic chemistry: background on the most basic definitions of chemistry, a bit of history, and highlighting why and how chemistry is important. The basic units in chemistry will be defined: atoms, molecules, subatomic particles. Then, we will discuss them from a beginner point of view, and formulate them in the format of questions.
Therefore, we aim this article to people that are unfamiliar with chemistry or with science in general. If you are a teacher, you might want to redirect your students here.
Disclaimer: This is not intended to be a comprehensive description of each concept, but rather an introduction to each of them: chemistry basics for beginners. We cite and include sources that we consider useful for expanding them further.
So withoutfurther ado, lets dive in!
Read Also: What Does Point Mean In Geometry
Get Free Full Access To:
- Flashcards
Take an atom. Prise it open and look inside. Of course, you cant really cut open an atom like that – they are far too small to be seen, apart from under the most powerful electron microscopes. But if you could, what would you see?
In the centre of the atom, you find a dense mass called the nucleus. Imagine 6 million cars squashed up together into a lump just 1 foot wide, 1 foot deep and 1 foot tall. Thats how dense the nucleus is. It contains protons, which are positively charged, and neutrons, which have a neutral charge.
Surrounding the nucleus, you find tiny negatively charged particles called electrons. Scientists like to imagine them as particles, but in reality they behave like waves at times – almost like a light wave. We imagine them whizzing around the nucleus in circular paths called orbitals. But although this sounds busy, the atom is mostly empty space. If an atom was the size of a cathedral, how big do you think its nucleus would be? The answer – the size of a fly.
The structure of an atom. The electrons, shown in red, orbit the nucleus, which contains a dense mass of protons and neutrons.commons.wikimedia.org
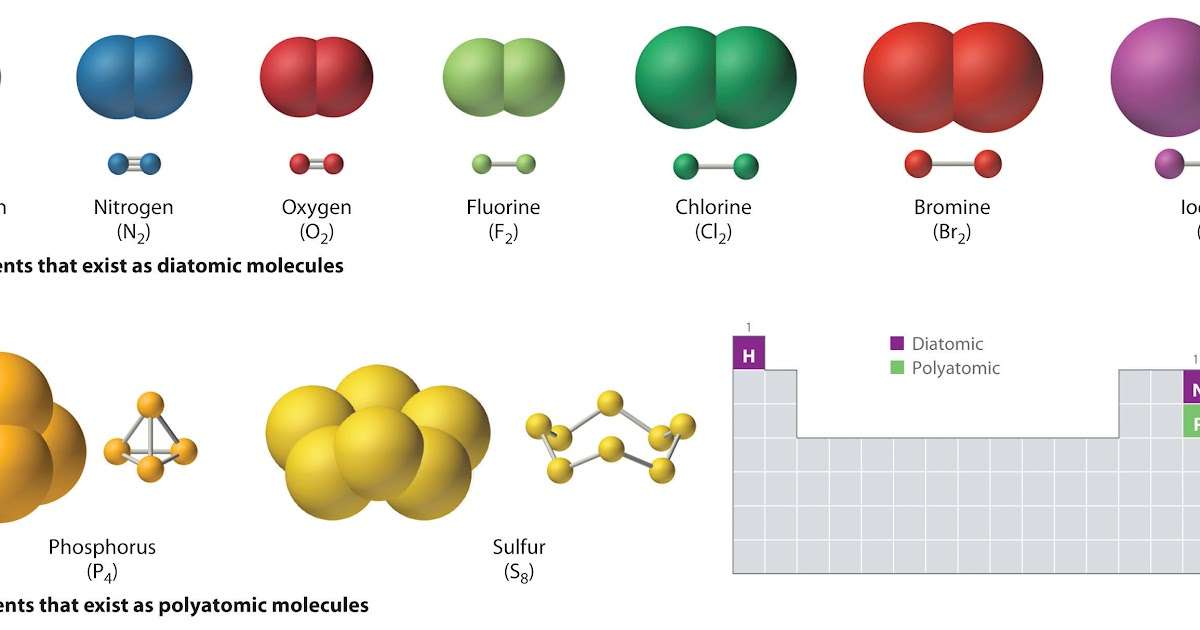
Left: a diatomic molecule. Right: a giant lattice.commons,wikimedia.org
This brings up questions: why do atoms lose or gain electrons? Why dont they move their protons about? What do lattices form and what forces hold them together? Why do some substances react so quickly and yet others are practically inert, meaning they dont react at all?
What Are The Branches Of Chemistry And Their Definition
Ernest Z. Jackie Shlecter Priya mrpauller.weebly.com
The five major branches of chemistry are organic, inorganic, analytical, physical, and biochemistry. These divide into many sub-branches.
Explanation:
ORGANIC CHEMISTRY
Organic chemistry involves the study of the structure, properties, and preparation of chemical compounds that consist primarily of carbon and hydrogen.
Organic chemistry overlaps with many areas including
- Medicinal chemistry the design, development, and synthesis of medicinal drugs. It overlaps with pharmacology .
- Organometallic chemistry the study of chemical compounds containing bonds between carbon and a metal.
- Polymer chemistry the study of the chemistry of polymers.
- Physical organic chemistry the study of the interrelationships between structure and reactivity in organic molecules.
- Stereochemistry the study of the spatial arrangements of atoms in molecules and their effects on the chemical and physical properties of substances.
INORGANIC CHEMISTRY
Inorganic chemistry is the study of the properties and behaviour of inorganic compounds.
It covers all chemical compounds except organic compounds.
Inorganic chemists study things such as crystal structures, minerals, metals, catalysts, and most elements in the Periodic Table.
Branches of inorganic chemistry include:
ANALYTICAL CHEMISTRY
Analytical chemistry involves the qualitative and quantitative determination of the chemical components of substances.
PHYSICAL CHEMISTRY
BIOCHEMISTRY
Recommended Reading: How To Find Distance In Physics
Why Do Two Chemical Compounds React
Chemistrystudies changes in matter. A chemicalreaction is a process in which one set of chemical compounds aretransformed into another. Reaction occur when there is an interaction betweenthe compounds in which some initial bonds are broken and some new bonds areformed.
Why does this happen? In simple terms, because the energy holding the new bonds together is higher than the energy that held the initial bonds. This is the definition of a thermodynamically favored process. Favorable thermodynamics is the most fundamental step that leads two compounds to react with each other. Other drastically important factor is reaction kinetics.
Kinetics And Rate Equations
Now that we know why atoms react from a chemical point of view, we can turn our attention back to physics. Kinetics is the study of the motion of particles and how this affects changing systems. Take iron and water, for example. They react to produce iron oxide. At room temperature, this reaction is very slow. But if you instead react the iron with steam, the reaction happens much faster. Why is this the case?
In Kinetics, youll learn about the rates of different reactions and how we can manipulate them. Heat is one way of increasing the speed of a reaction, but youll also explore other factors such as surface area and concentration. Youll expand this knowledge in Rate Equations. By the end of the topic, youll know how to work out the rate of reactions both experimentally and theoretically.
Also Check: Glencoe Algebra 1 Chapter 6 Answer Key
When Was Chemistry Discovered
Very simple chemical processes were performed even during ancient history, at 1000 BC, much before any basic chemistry concepts or laws were actually established. Extraction of metals from ore or getting compounds out of natural sources such as plants, are examples of chemistry that was first performed thousands of years ago and are still a thing today.
Alchemy is what we call the protoscience of chemistry.
The Alchemist in Search of the Philosophers Stone
Alchemytried to explain the nature, properties and transformations of matter. But itwas not science, but rather a set of myths and magic. It is agreed upon that,the transition from alchemy to modern chemistry as we know it, started on the17th century, with the publication of The Sceptical Chymist by Robert Boyle , who isconsidered the father of modern chemistry. The difference between chemistry andalchemy is the application of the scientific method.
List Of Basic Chemistry Element Terms And Definitions
A Branch of science which deals with the structure,properties constituents and change which takes place is known as chemistry.Origin of chemistry -: Chemes means black color. Egyptian called black earth as chemi and in early age the study of chemical science was known as chemistry.Lavoisier is known as father of Modern Chemistry.Here List of Basic Chemistry Element Terms and Definitions-
Read Also: Geometry Circles Chapter Test Review
Questions: Fill In The Blanks
Solution.
What Types Of Chemical Compounds Are There
There are three basic types of chemical compounds, and should be briefly introduced in this post about basic chemistry concepts. All of them are the result of bonding atoms together. The difference is in the nature of the forces that hold together those atoms.
- In molecules , which are neutral compounds of individual nature, atoms are glued together by covalent bonds. Covalent bonds generally occur between two non-metal atoms, which share pairs of electrons, or bonding pairs.
- In ionic compounds, atoms are in ionic form and are held together by ionic forces, giving rise to large networks of oppositely charged ions. Ionic bonds occur between metals and non-metals.
- When extended networks of atoms are formed between one or more types of metal atoms, we are talking about metallic bonds.
Don’t Miss: What Is Estimate In Math