The Complexities Of Biochemistry
Protein is a foundational material in the structure of most living things, and as such it is rather like concrete or steel. Just as concrete is a mixture of other ingredients and steel is an alloy of iron and carbon, proteins, too, are made of something more basic: amino acids. These are organic compounds made of carbon, hydrogen, oxygen, nitrogen, and sulfur bonded in characteristic formations.
Amino acids are discussed in more depth within the essay devoted to that topic, though, as noted in that essay, it is impossible to treat such a subject thoroughly without going into an extraordinarily lengthy and technical discussion. Such is the case with many topics in biochemistry, the area of the biological sciences concerned with the chemical substances and processes in organisms: the deeper within the structure of things one goes, and the smaller the items under investigation, the more complex are the properties and interactions.
What Are Protein Families
Allproteins bind to other molecules in order to complete their tasks, and theprecise function of a protein depends on the way its exposed surfaces interactwith those molecules. Proteins with related shapes tend to interact withcertain molecules in similar ways, and these proteins are therefore considereda protein family. The proteinswithin a particular family tend to perform similar functions within the cell.
Proteinsfrom the same family also often have long stretches of similar amino acidsequences within their primary structure. These stretches have been conserved throughevolution and are vital to the catalytic function of the protein. For example,cell receptor proteins contain different amino acid sequences at their bindingsites, which receive chemical signals from outside the cell, but they are moresimilar in amino acid sequences that interact with common intracellularsignaling proteins. Protein families may have many members, and they likelyevolved from ancient gene duplications. These duplications led to modificationsof protein functions and expanded the functional repertoire of organisms overtime.
Does Kidney Damage Go Away
In general, it does not. The goal is to detect kidney disease and damage early to minimize the damage and prolong kidney function. If the proteinuria detected is due to a kidney infection or urinary tract infection, the kidneys frequently will return to normal function as the infection resolves. If it is due to a medication, then in most cases the kidneys are likely to return to normal or near-normal function when the medication is stopped.
Recommended Reading: Age Word Problems With Solutions
Peptide Bond Formation And Primary Protein Structure
Within cellular systems, proteins are linked together by a large enzyme complex that contains a mixture of RNA and proteins. This complex is called the ribosome. Thus, as the amino acids are linked together to form a specific protein, they are placed within a very specific order that is dictated by the genetic information contained within the messenger RNA molecule. This specific ordering of amino acids is known as the proteins primary sequence. The translation mechanism used by the ribosome to synthesize proteins will be discussed in detail in Chapter XX. This chapter will focus only on the chemical reaction occurring during synthesis and the physical properties of the resulting peptides/proteins.
The primary sequence of a protein is linked together using dehydration synthesis that combine the carboxylic acid of the upstream amino acid with the amine functional group of the downstream amino acid to form an amide linkage . Similarly, the reverse reaction is hydrolysis and requires the incorporation of a water molecule to separate two amino acids and break the amide bond. Notably, the ribosomeserves as the enzyme that mediates the dehydration synthesis reactions required to build protein molecules, whereas a class of enzymes called proteases are required for protein hydrolysis.
Figure 2.10 Formation of the Peptide Bond. The addition of two amino acids to form a peptide requires dehydration synthesis.
Image fromV.K. Chang
Image by: CNX OpenStax via Wikimedia Commons
Aerobic Exercise Protein Needs
Endurance athletes differ from strength-building athletes in that endurance athletes do not build as much muscle mass from training as strength-building athletes do. Research suggests that individuals performing endurance activity require more protein intake than sedentary individuals so that muscles broken down during endurance workouts can be repaired. Although the protein requirement for athletes still remains controversial , research does show that endurance athletes can benefit from increasing protein intake because the type of exercise endurance athletes participate in still alters the protein metabolism pathway. The overall protein requirement increases because of amino acid oxidation in endurance-trained athletes. Endurance athletes who exercise over a long period use protein as a source of 510% of their total energy expended. Therefore, a slight increase in protein intake may be beneficial to endurance athletes by replacing the protein lost in energy expenditure and protein lost in repairing muscles. One review concluded that endurance athletes may increase daily protein intake to a maximum of 1.21.4 g per kg body weight.
Recommended Reading: Evaluating Functions Worksheet Algebra 2 Answer Key
Quaternary Structure Of Protein
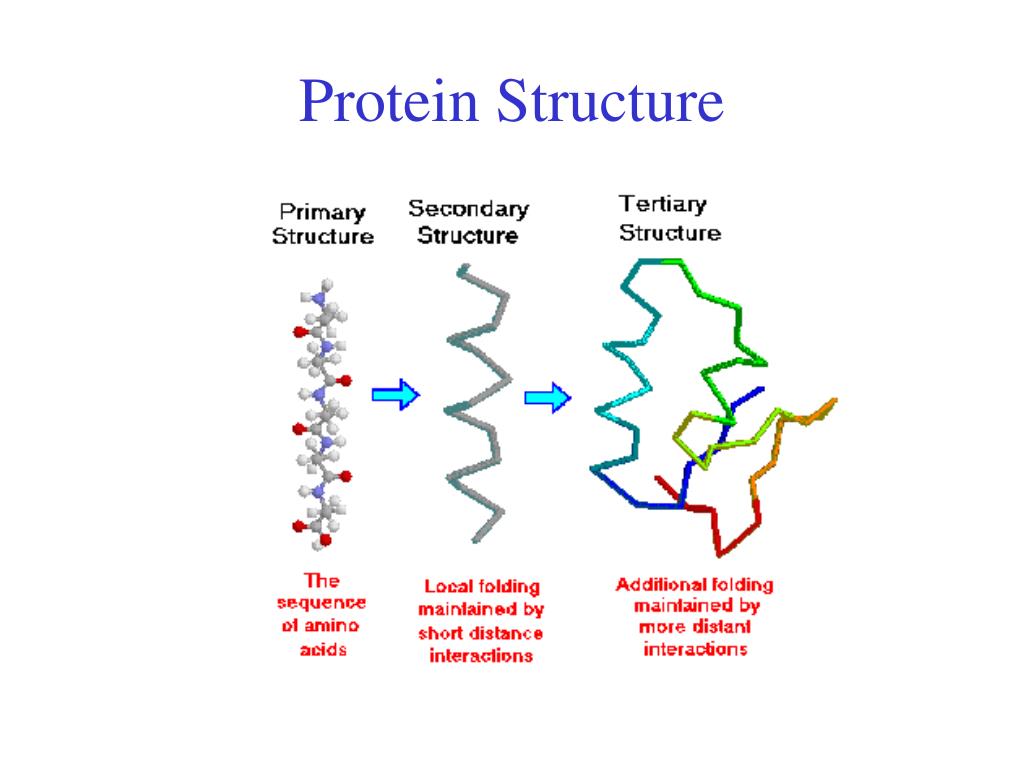
The spatial arrangement of various tertiary structures gives rise to the quaternary structure. Some of the proteins are composed of two or more polypeptide chains referred to as sub-units. The spatial arrangement of these subunits with respect to each other is known as quaternary structure.
Quaternary Structure of Protein
The exact amino acid sequence of each protein drives it to fold into its own unique and biologically active three-dimensional fold also known as the tertiary structure. Proteins consist of different combinations of secondary elements some of which are simple whereas others are more complex. Parts of the protein chain, which have their own three-dimensional fold and can be attributed to some function are called domains. These are considered today as the evolutionary and functional building blocks of proteins.
Many proteins most of which are enzymes contain organic or elemental components needed for their activity and stability. Thus the study of protein evolution not only gives structural insight but also connects proteins of quite different parts of the metabolism.
If I Have A Positive Urine Protein Test What Other Tests Might Be Done In Follow Up
A positive urine protein test tells your healthcare practitioner that protein is present in your urine, but it does not indicate which types are present or the cause of the proteinuria. When investigating the reason, your healthcare practitioner may order additional laboratory tests, such as:
- Comprehensive metabolic panel includes tests such as BUN, creatinine, albumin and total protein, which help evaluate kidney and liver function
- A full urinalysisif a condition such as urinary tract infection is also suspected
- Serum and urine protein electrophoresis testto determine which proteins are being released into the urine and in what quantities this is especially true if your healthcare practitioner suspects abnormal monoclonal protein production, such as with multiple myeloma or lymphoma.
See the article on Proteinuria for more on follow-up tests.
If kidney disease or damage is suspected, your healthcare practitioner may also order imaging scans to evaluate the appearance of your kidneys.
Read Also: Geometry Seeing Doing Understanding Answer Key Pdf
Inaccuracy Of Crude Protein
There’s some evidence that the facts listed on menus don’t accurately report the actual nutrient — including protein — content of the food. One study, published in Appetite in 2015, found that the actual protein content of restaurant food offered on a college campus differed significantly from the nutrition info posted on the menu. In addition, the real protein content of milk can differ from the amount of crude protein, according to a report from Cornell University. This difference can have a significant effect on making dairy products, like cheese, notes the report.
Cell Signaling And Ligand Binding
Many proteins are involved in the process of cell signaling and signal transduction. Some proteins, such as insulin, are extracellular proteins that transmit a signal from the cell in which they were synthesized to other cells in distant tissues. Others are membrane proteins that act as receptors whose main function is to bind a signaling molecule and induce a biochemical response in the cell. Many receptors have a binding site exposed on the cell surface and an effector domain within the cell, which may have enzymatic activity or may undergo a conformational change detected by other proteins within the cell.:25181
Antibodies are protein components of an adaptive immune system whose main function is to bind antigens, or foreign substances in the body, and target them for destruction. Antibodies can be secreted into the extracellular environment or anchored in the membranes of specialized B cells known as plasma cells. Whereas enzymes are limited in their binding affinity for their substrates by the necessity of conducting their reaction, antibodies have no such constraints. An antibody’s binding affinity to its target is extraordinarily high.:27550
Recommended Reading: What Is The Molecular Geometry Of Ccl4
Influence Of Heat Treatment On Milk Proteins
The caseins are stable to heat treatment. Typical high temperature short time pasteurization conditions will not affect the functional and nutritional properties of the casein proteins. High temperature treatments can cause interactions between casein and whey proteins that affect the functional but not the nutritional properties. For example, at high temperatures, ß-lactoglobulin can form a layer over the casein micelle that prevents curd formation in cheese.
The whey proteins are more sensitive to heat than the caseins. HTST pasteurization will not affect the nutritional and functional properties of the whey proteins. Higher heat treatments may cause denaturation of ß-lactoglobulin, which is an advantage in the production of some foods and ingredients because of the ability of the proteins to bind more water. Denaturation causes a change in the physical structure of proteins, but generally does not affect the amino acid composition and thus the nutritional properties. Severe heat treatments such as ultra high pasteurization may cause some damage to heat sensitive amino acids and slightly decrease the nutritional content of the milk. The whey protein -lactalbumin, however, is very heat stable.
Cellular And Tissue Provisioning
Protein is an essential component for every type of cell in the body, including muscles, bones, organs, tendons, and ligaments. Protein is also needed in the formation of enzymes, antibodies , hormones, blood-clotting factors, and blood-transport proteins. The body is constantly undergoing renewal and repair of tissues. The amount of protein needed to build new tissue or maintain structure and function depends on the rate of renewal or the stage of growth and development . For example, the intestinal tract is renewed every couple of days, whereas blood cells have a life span of 60 to 120 days. Furthermore, an infant will utilize as much as one-third of the dietary protein for the purpose of building new connective and muscle tissues.
Also Check: Abiotic Meaning In Science
The Chemistry Of Amino Acids
IntroductionWhy learn this?
Amino acids play central roles both as building blocks of proteins and as intermediates in metabolism. The 20 amino acids that are found within proteins convey a vast array of chemical versatility. The precise amino acid content, and the sequence of those amino acids, of a specific protein, is determined by the sequence of the bases in the gene that encodes that protein. The chemical properties of the amino acids of proteins determine the biological activity of the protein. Proteins not only catalyze all of the reactions in living cells, they control virtually all cellular process. In addition, proteins contain within their amino acid sequences the necessary information to determine how that protein will fold into a three dimensional structure, and the stability of the resulting structure. The field of protein folding and stability has been a critically important area of research for years, and remains today one of the great unsolved mysteries. It is, however, being actively investigated, and progress is being made every day.
As we learn about amino acids, it is important to keep in mind that one of the more important reasons to understand amino acid structure and properties is to be able to understand protein structure and properties. We will see that the vastly complex characteristics of even a small, relatively simple, protein are a composite of the properties of the amino acids which comprise the protein.
Signal Sequences In Protein Synthesis
Protein must be delivered to the proper destination in the cell to function properly. Signal sequences within the protein itself act like “zip codes” to ensure correct delivery. The synthesis of secreted proteins like insulin and of proteins that will be integral to the plasma membrane occurs at a ribo-some tethered to the endoplasmic reticulum , which is a system of membranes that transport materials within cells. The peptides formed there are then translocated into the lumen , or channel, of the endoplasmic reticulum, where they will be formed into a polypeptide chain. This translocation occurs because of a specific signal sequence that is formed by the first twenty or so amino acids in the protein. The core of this sequence consists of ten to fifteen amino acids that have hydrophobic side chains such as alanine, leucine, valine, isoleucine, and phenylalanine, which are usually cleaved from the protein later on. The nascent polypeptide chain is guided along this path by a signal receptor protein.
You May Like: What Does K Mean In Physics
Red And Processed Meat And Cancer Risk
Protein is essential for good health, but some high protein foods may be better for our health than others. In particular, consuming high amounts of red and processed meat has been associated with an increased risk of certain cancers.8Red meat is a good source of protein as well as many other essential nutrients such as iron, vitamin B12, and zinc, and does not necessarily need to be avoided altogether to reduce risk. The World Cancer Research Fund recommends we try to consume no more than three portions of red meat per week and very little, if any, processed meat.8
What Is The Longest Chemical Formula
longest chemical
It has a theoretical isoelectric point of 6.01. The protein’s empirical chemical formula is C169,719H270,466N45,688O52,238S911. It has a theoretical instability index of 42.41, classifying the protein as unstable.
Also Know, what is the chemical name of Titin the largest known protein? isoleucine” , which is the “chemical name of titin, the largest known protein.” Also, there’s some dispute about whether this is really a word.
Secondly, what is the most complicated chemical formula?
Maitotoxin is one of the most complex organic compounds ever characterized, from a living organism. It is a toxic polyether compound with a very, very low lethal dosage, in the order of nanograms for mice.
What is the chemical name of io7?
Iodine oxide
Read Also: Ccl4 Resonance Structures
What Is Being Tested
Urine protein tests detect and/or measure protein being released into the urine. Normal urine protein elimination is less than 150 mg/day and less than 30 mg of albumin/day. Elevated levels may be seen temporarily with conditions such as infections, stress, pregnancy, diet, cold exposure, or heavy exercise. Persistent protein in the urine suggests possible kidney damage or some other condition that requires additional testing to
Urine protein tests detect and/or measure protein being released into the urine. Normal urine protein elimination is less than 150 mg/day and less than 30 mg of albumin/day. Elevated levels may be seen temporarily with conditions such as infections, stress, pregnancy, diet, cold exposure, or heavy exercise. Persistent protein in the urine suggests possible kidney damage or some other condition that requires additional testing to determine the cause.
There are several different kinds of urine protein tests, including:
- A semi-quantitative protein dipstick may be performed as part of a urinalysis, generally on a random urine sample.
- The quantity of protein in a 24-hour urine sample may be measured and reported as the amount of protein released per 24 hours.
- The amount of protein in a random urine sample may be measured along with urine creatinine and reported as the ratio of urine protein to creatinine .
Secondary Structure Of Protein
- The proteins do not exist in just simple chains of polypeptides.
- These polypeptide chains usually fold due to the interaction between the amine and carboxyl group of the peptide link.
- The structure refers to the shape in which a long polypeptide chain can exist.
- They are found to exist in two different types of structures helix and pleated sheet structures.
- This structure arises due to the regular folding of the backbone of the polypeptide chain due to hydrogen bonding between -CO group and -NH groups of the peptide bond.
- However, segments of the protein chain may acquire their own local fold, which is much simpler and usually takes the shape of a spiral an extended shape or a loop. These local folds are termed secondary elements and form the proteins secondary structure.
Secondary Structure of Protein
Also Check: Eoc Fsa Practice Test Algebra 1 Calculator Portion
Chapter 3 Proteins And Amino Acids
Proteins are complex, organic compounds composed of many amino acids linked together through peptide bonds and cross-linked between chains by sulfhydryl bonds, hydrogen bonds and van der Waals forces. There is a greater diversity of chemical composition in proteins than in any other group of biologically active compounds. The proteins in the various animal and plant cells confer on these tissues their biological specificity.
What Gets Stored In A Cookie
This site stores nothing other than an automatically generated session ID in the cookie no other information is captured.
In general, only the information that you provide, or the choices you make while visiting a web site, can be stored in a cookie. For example, the site cannot determine your email name unless you choose to type it. Allowing a website to create a cookie does not give that or any other site access to the rest of your computer, and only the site that created the cookie can read it.
Read Also: Ccl4 Valence Electrons