Electron Geometry Vs Molecular Geometry: Whats The Difference
Interesting and intriguing chemistry is always fun! While understanding what matter is made of, we learn about so many new things that we simply lose ourselves in the beautiful world of chemistry.
However, a few concepts can be slightly difficult to comprehend because they seem similar or because they are just confusing! One such concept is the difference between electron geometry and molecular geometry. To help you out, we are shedding light on electron geometry vs molecular geometry in this article.
Electron geometry teaches us about the arrangement of different electron groups. Molecular geometry, on the other hand, helps us understand the entire atom and its arrangement. It is the 3D arrangement of all the atoms in a particular molecule.
So, when you compare them, you will note that atoms have different arrangements in electron geometry and molecular geometry.
Difference Between Molecular Geometry And Electron Geometry
| Molecular geometry | Electron geometry |
| Molecular Geometry is the arrangement of atoms in a molecule, normally relative to a single central atom. | Electron Geometry is the arrangement of electron pairs around a central atom. |
| It excludes lone pairs in deciding the shape of a molecule, although repulsion from lone pair is taken into account only in bond angles. | It considers the presence of both bond pair and lone pair of electrons in determining the shape. |
The Influence Of Thermal Excitation
Since the motions of the atoms in a molecule are determined by quantum mechanics, “motion” must be defined in a quantum mechanical way. The overall quantum mechanical motions translation and rotation hardly change the geometry of the molecule. In addition to translation and rotation, a third type of motion is molecular vibration, which corresponds to internal motions of the atoms such as bond stretching and bond angle variation. The molecular vibrations are harmonic , and the atoms oscillate about their equilibrium positions, even at the absolute zero of temperature. At absolute zero all atoms are in their vibrational ground state and show zero point quantum mechanical motion, so that the wavefunction of a single vibrational mode is not a sharp peak, but an exponential of finite width . At higher temperatures the vibrational modes may be thermally excited , but they oscillate still around the recognizable geometry of the molecule.
To get a feeling for the probability that the vibration of molecule may be thermally excited,we inspect the Boltzmann factorβ â¡ exp, where ÎE is the excitation energy of the vibrational mode, k the Boltzmann constant and T the absolute temperature. At 298 K , typical values for the Boltzmann factor β are:
- β = 0.0890 for ÎE = 0500 cmâ1
- β = 0.0080 for ÎE = 1000 cmâ1
- β = 0.0007 for ÎE = 1500 cmâ1.
Read Also: Reversible Figure-ground Relationship
What Is Molecular Geometry
Molecular geometry is the shape of a molecule predicted by considering only bond electron pairs. In this case, lone electron pairs are not taken into account. Moreover, double bonds and triple bonds are considered as single bonds. The geometries are determined based on the fact that lone electron pairs need more space than bonding electron pairs. For example, if a certain molecule is composed of two pairs of bonding electrons along with a lone pair, the molecular geometry is not linear. The geometry there is bent or angular because the lone electron pair needs more space than two bonding electron pair.
Determination Of Electron Geometry
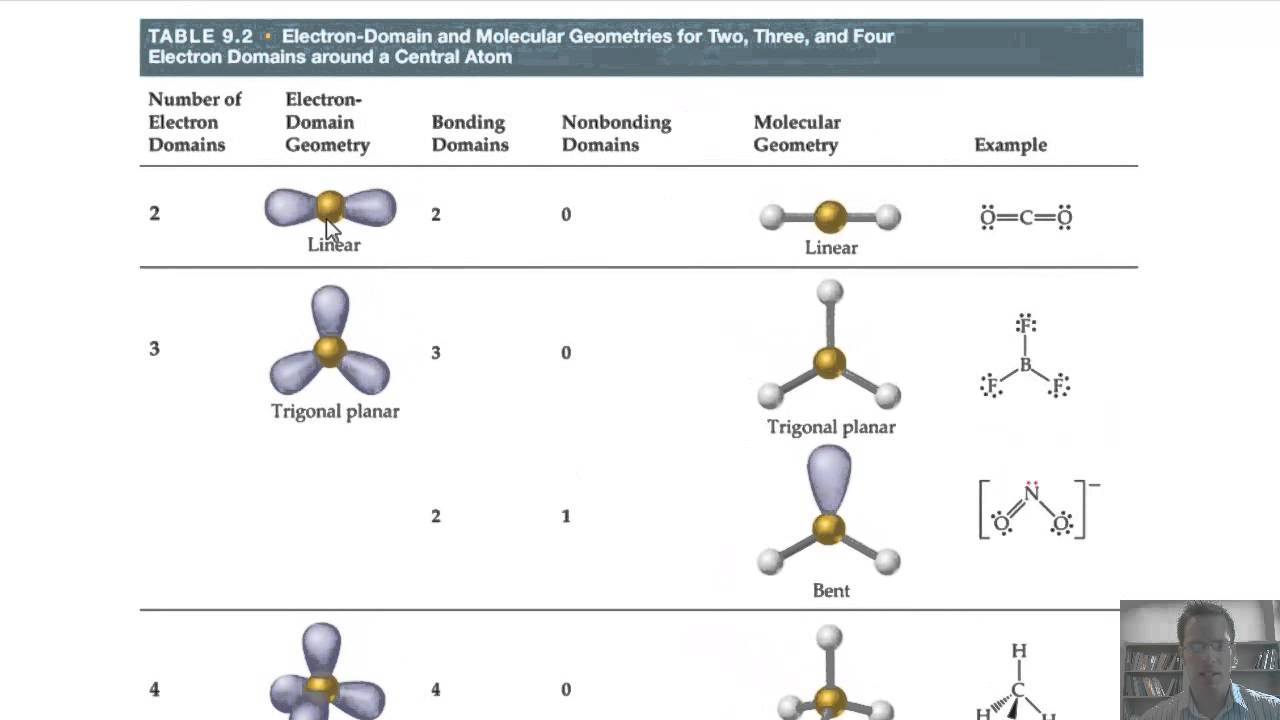
Since electrons are always moving and their paths cannot be accurately figured, the arrangement of electrons is described in terms of electron density distribution.
Electron geometry is determined by the number of electron pairs. The following table gives an idea of electronic geometry according to the number of electron pairs.
Don’t Miss: Exponent Rules Review Worksheet
Types Of Molecular Structure
A bond angle is the geometric angle between two adjacent bonds. Some common shapes of simple molecules include:
The bond angles in the table below are ideal angles from the simple VSEPR theory , followed by the actual angle for the example given in the following column where this differs. For many cases, such as trigonal pyramidal and bent, the actual angle for the example differs from the ideal angle, and examples differ by different amounts. For example, the angle in H2S differs from the tetrahedral angle by much more than the angle for H2O does.
| Atoms bonded to |
|---|
This Problem Has Been Solved
VSEPR theory includes two typesof geometries: electron geometry and molecular geometry. What isthe difference between the two geometries?
| -Electron geometry describes the arrangement of the electrongroups in a molecule, whereas the molecular geometrydescribes the arrangement of the atoms. |
| -There is no difference between electron and moleculargeometries they are simply different names for the samething. |
| Lone pairs are included in the electron geometry, but play norole in the molecular geometry. |
| -The electron geometry only considers lone pairs of electronsand the molecular geometry only considers the atoms. |
Read Also: Exponential Growth And Decay Worksheet Algebra 1
Also Check: What Is The Molecular Geometry Of Ccl4
Main Differences Between Electron Geometry And Molecular Geometry
H2o2 Lewis Structure Molecular Geometry Hybridization And Polarity
H2O2 is a chemical compound with the IUPAC name Hydrogen Peroxide. It is the simplest peroxide compound, i.e., a molecule containing an Oxygen-Oxygen single bond.
It is a pale blue liquid in its standard state and slowly reacts with sunlight and decomposes into water and oxygen.
2H2O2 2H2O + O2
This is an example of a disproportionation reaction as O is a -1 state on the left-hand side while it exists in -2 and +0 oxidation states on the right-hand side.
H2O2 has a melting point of -0.43 Celsius . A low melting point indicates the tendency of the compound to remain in the liquid state.
It also exhibits a relatively high boiling point of 150.2 Celsius , attributed to strong hydrogen bonding interactions with water and other H2O2 molecules.
Industrially, H2O2 is prepared using Anthraquinone as a catalyst. The reaction is as follows :
2H2O + O2 2H2O2
This is an example of a comproportionation reaction, i.e., the reverse of a disproportionation reaction.
It is a strong oxidizing agent, and hence, it finds wide applications as a bleaching agent and disinfectant.
It also plays a vital role in organic synthesis, used in the oxidation/reduction of various functional groups. It is also used as an oxidizer in spacecraft since oxygen is not available for combustion in outer space.
H2O2 is a toxic byproduct of several biological processes, and several enzymatic reactions are dedicated to the decomposition of H2O2.
It is toxic and corrosive and hence must be handled with caution.
You May Like: What Is The Molecular Geometry Of Ccl4
Clf3 Molecular Geometry Or Shape
You can also use the AXN method to determine the molecular geometry or electron geometry of ClF3.
- A represents the central atom.
- X represents the bonded atoms to the central atom.
- N represents the lone pairs on the central atom
With the help of the ClF3 Lewis dot structure, we know chlorine is the central atom that contains 2 lone pairs and is attached to 3 bonded atoms.
So, the ClF3 formula becomes AX3N2.
According to the AX3N2 formula, ClF3 molecular geometry is T-shaped and electron geometry is trigonal pyramidal.
| Bonded atoms |
| Trigonal bipyramidal |
VSEPR Chart
As ClF3 has 2 lone pair or 3 bond repulsion units and it formed T-shaped or trigonal pyramidal geometry, Its FClF involving the axial atoms bond angle is 175º and FClF involving the one axial atom and one equatorial bond angle is around 90º.
You May Like: What Is The Molecular Geometry Of Ccl4
Main Difference Electron Geometry Vs Molecular Geometry
The geometry of a molecule determines the reactivity, polarity and biological activity of that molecule. The geometry of a molecule can be given as either the electron geometry or the molecular geometry. The VSEPR theory can be used to determine the geometries of molecules. Electron geometry includes the lone electron pairs present in a molecule. Molecular geometry can be determined by the number of bonds that a particular molecule has. The main difference between electron geometry and molecular geometry is that electron geometry is found by taking both lone electron pairs and bonds in a molecule whereas molecular geometry is found using only the bonds present in the molecule.
Also Check: Paris Jackson Biological
Chapter 6 Molecular Structure Of Compounds
- Predict the structures of small molecules using valence shell electron pair repulsion theory
Thus far, we have used two-dimensional Lewis structures to represent molecules. However, molecular structure is actually three-dimensional, and it is important to be able to describe molecular bonds in terms of their distances, angles, and relative arrangements in space . A bond angle is the angle between any two bonds that include a common atom, usually measured in degrees. A bond distance is the distance between the nuclei of two bonded atoms along the straight line joining the nuclei. Bond distances are measured in Ångstroms or picometers .
Figure 1. Bond distances and angles are shown for the formaldehyde molecule, H2CO.
Is Electron Domain Geometry And Molecular Geometry
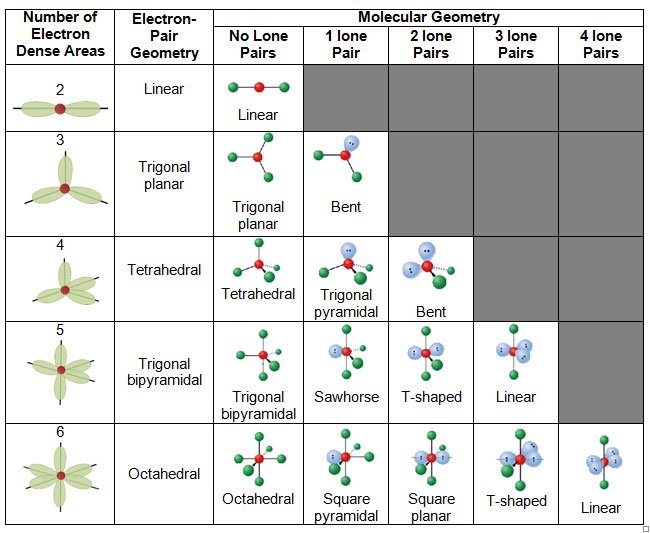
electronElectron domain geometrieselectronmolecular geometriesmolecule
Electron geometry teaches us about the arrangement of different electron groups. Molecular geometry, on the other hand, helps us understand the entire atom and its arrangement. It is the 3D arrangement of all the atoms in a particular molecule.
Also Know, why is so2 bent and not linear? CO2 is linear and the SO2 structure is bent, because CO2 has a negative oxygen on each side of the positive carbon they cancel each other out. In the SO2 structure the oxygen are not in lined with each other which means there a positive and negative end.
One may also ask, how do you find the electron domain and molecular geometry?
Count the total number of electron domains. Use the angular arrangement of the chemical bonds between the atoms to determine the molecular geometry. Keep in mind, multiple bonds count as one electron domain. In other words, a double bond is one domain, not two.
What is the molecular geometry of h2o?
Water or H2O has 8 electrons around the central oxygen atom. This means there are four electron pairs arranged in a tetrahedral shape. There are two bonding pairs and two lone pairs. The resulting shape is bent with an H-O-H angle of 104.5°.
Recommended Reading: Eoc Fsa Warm Ups Algebra 1 Answers
What Is Electrongeometry
Electron geometry is the shape of a molecule predicted by considering both bond electron pairs and lone electron pairs. The VSEPR theory states that electron pairs located around a certain atom repel each other. These electron pairs can be either bonding electrons or non-bonding electrons.
The electron geometry gives the spatial arrangement of all the bonds and lone pairs of a molecule. The electron geometry can be obtained using VSEPR theory.
Key Concepts And Summary
VSEPR theory predicts the three-dimensional arrangement of atoms in a molecule. It states that valence electrons will assume an electron-pair geometry that minimizes repulsions between areas of high electron density . Molecular structure, which refers only to the placement of atoms in a molecule and not the electrons, is equivalent to electron-pair geometry only when there are no lone electron pairs around the central atom.
Don’t Miss: Fsa Algebra 1 Eoc Answers
What’s The Main Difference Between Electron Pair Geometry & Molecular Geometry
Molecular geometry in chemistry is a concept that refers to the arrangement of atoms about the central atom in a three-dimensional space.
The definition of electron geometry is the arrangement about electron groups of atoms. The bond is 105 degrees.
If the lone pairs of electrons are situated in the molecule, it changes the molecular geometry and not the electron pairs.
When electrons are bonded to the central atom and do not have lone pairs, the electron geometry and molecular geometry are similar.
Molecular Geometry Of Nh4
While the Lewis Structure is a 2-dimensional depiction of an atom of a molecule, molecular geometry is the visualization and designing of the atoms in a 3-dimensional space.
The concept of molecular geometry aims to depict the generic shape and structure of a molecule, accurate to the length between different bonds, the bond and torsional angles, other geometrical factors and variables that govern the shape and arrangement of an atom, and therefore, a molecule.
Molecular geometry also helps to determine the atomic properties of an element, such as polarity, magnetism, reactivity, color, biological potency, and 3-dimensional space alignment.
The properties of an atom identified through molecular geometry help in understanding the behavior, utility, and reactivity of the element.
Depending upon their geometry, various molecular structures can be classified into linear, angular, trigonal planar, octahedral, trigonal pyramidal, among others.
One can draw the 3-dimensional structure of an atom once they have the Lewis Structure of an atom.
The 3-dimensional geometrical structure of ammonium, NH4+ is referred to as Tetrahedral. Nitrogen, having 5 valence shell electrons, along with 4 from Hydrogen, should have had 9 electrons.
But the + sign decrees that NH4+ has 8 valence shell electrons, due to the positive ion. This means that NH4+ has 4 pairs in total, which are bonded due to the 4 atoms of hydrogen.
Below is the image of the geometrical representation of the NH4+ molecule.
Read Also: Movement Geography Example
S Used To Find The Shape Of The Molecule
To sum up there are four simple steps to apply the VSEPR theory.
What Is Electron Geometry
Electron geometry is the shape of a molecule predicted by considering both bond electron pairs and lone electron pairs. The VSEPR theory states that electron pairs located around a certain atom repel each other. These electron pairs can be either bonding electrons or non-bonding electrons.
The electron geometry gives the spatial arrangement of all the bonds and lone pairs of a molecule. The electron geometry can be obtained using VSEPR theory.
Read Also: What Does Abiotic Mean In Biology
Determination Of Molecular Geometry
The shape of a molecule is determined by the bonded atom, although this does not mean the shape itself is unaffected by the presence of lone pair.
Molecular geometry includes geometrical parameters such as bond lengths, bond angles, and torsional angles that help determine the position of atoms as well as a molecules general shape.
It influences a substances properties such as its reactivity, color, polarity, magnetism, biological activity, and phase of matter.
There are various techniques to determine molecular geometry such as Raman spectroscopy, infrared spectroscopy, and microwave spectroscopy.
What You Need Toknow About Electron Geometry
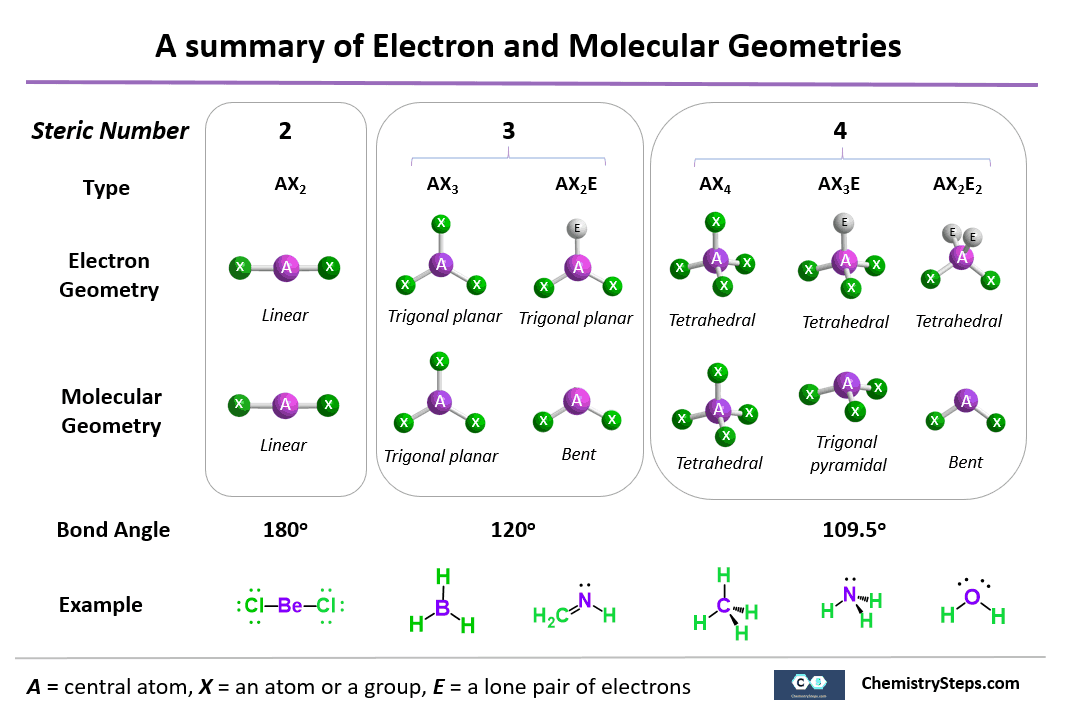
- Electron geometry is the geometry in which theshape of a molecule is predicted by considering both bond electron pairs andlone electron pairs.
- Electron geometry considers electron pair.
- Electron geometry helps give the arrangement ofelectron pair.
- Electron geometry includes both bonding andnon-bonding electron.
- Electron geometry can also be determined by theVESPR Theory.
- In electron geometry, number of total electronpairs is calculated.
Read Also: Unit 1 Test Geometry Basics Answers Key
What Is The Difference Between Electronic And Molecular Geometries
See Below
Explanation:
Electron Geometry is the shape the electrons take around the central atom. These electrons can be in either a chemical bond or in lone pairs.- Linear
Molecular GeometryThis is the shape the actual connections between atoms take in a compound. The shape is dictated by the electron geometry, though. The electron geometry is the scaffold, and the connections between the atoms depend on this scaffold.
Ammonia has electron geometry that is tetrahedral . It has a lone pair, though, and if you put this lone pair on top, then the connections between N-H make the NH3 compound look like a camera on a tripod this is called Trigonal Pyramidal and this is its molecular geometry.
What Is The Lewis Structure Of Nh4
As mentioned earlier, NH4+ is made up of Nitrogen and Hydrogen. If we look towards the periodic table, we will find Hydrogen in group 1.
This means that Hydrogen has 1 electron. NH4+ has 4 hydrogen atoms, therefore, there are 4 hydrogen electrons.
Nitrogens valence electron count, however, is 5, owing to its position in the 5th group of the periodic table. The plus sign denotes the absence of 1 electron therefore, it is minus one.
If we total out the number of electrons, it will be + 1 = 4 + 5 1 = 8.
We thus have 8 valence electrons here.
Keeping Nitrogen in the center, and considering Hydrogens position on the outside, we can place the 4 hydrogen atoms surrounding the single nitrogen atom.
Next is putting the chemical bonds.
Since the NH4+ atom has 8 valence electrons, our arrangement will be according to 2,4,6, and 8.
Referring to the octet rule, hydrogen needs only 2 valence electrons, which it already has. Even Nitrogen, which needs 8 electrons in the valence shell has all 8 of them, thereby forming a full exterior shell.
The loss of an electron is depicted by putting a + sign enclosing the Lewis structure.
Read Also: Paris Jackson Biological Parents