Phagocytosis Of Bacteria By Professional Phagocytes
Unlike phagocytosis of apoptotic cells where both professional and non-professional phagocytes may be involved, infection by various invading/ pathogenic microorganisms such as bacteria and viruses triggers immune responses where only professional phagocytes are involved.
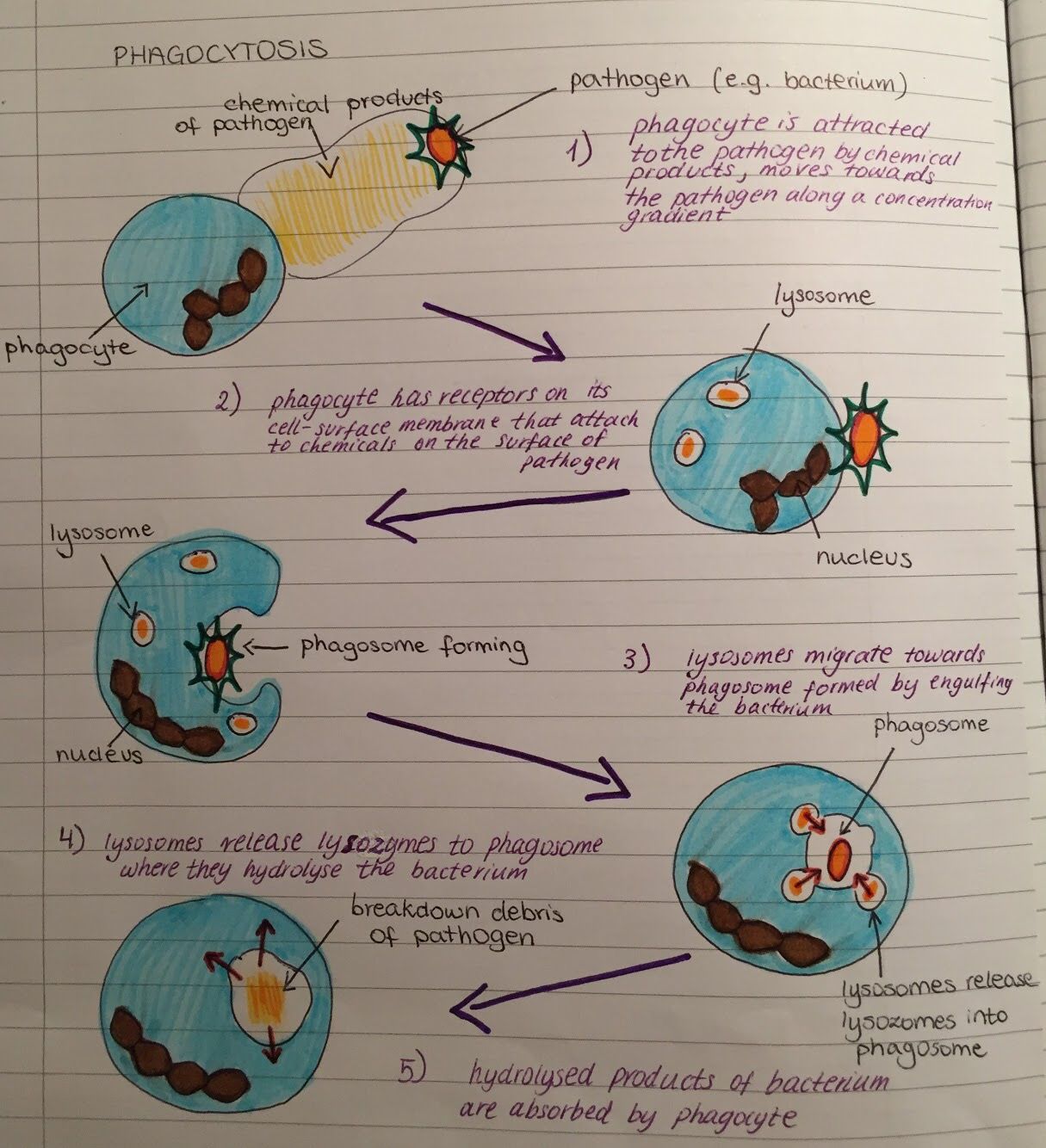
The following are steps through which phagocytic cells ingest bacteria:
Step 1: Activation
Infection of a given site in the body stimulates the delivery of neutrophils to the site through chemotaxis. Here, such attractants as tissue debris and bacteria product activate the movement of the phagocytic cells to move to the infected site.
* Neutrophils are able to reach the site fast because they are always circulating in the blood. Through such processes, these cells are able to migrate through the vascular walls to reach the target site.
Step 2: Adherence/Binding
Bacteria with such molecules/ substances as IgG or C3b are easily identified by the phagocytes. Here, such receptors as opsonin receptors on the plasma membrane of the phagocyte bind to these molecules allowing for the bacteria to be identified and ultimately ingested.
Here, it is worth noting that different types of phagocytes have different surface receptors. Therefore, it’s only through successful binding with the appropriate surface molecules on the invader that the organism can be ingested.
Step 3: Ingestion/Engulfment
Step 4: Enzyme action
Detection Of The Target Particle
The first phase in phagocytosis is the detection of the target particle. Detection is mediated by dedicated receptors on phagocytic cells. Receptors directly recognizing pathogen-associated molecular patterns are the pattern-recognition receptors . Some of these PRRs can initiate phagocytosis and thus constitute the non-opsonic receptors for phagocytosis. Other PRRs, for example TLRs, can bind to PAMPs but not induce phagocytosis. These receptors however, can prepare the cell for phagocytosis. Foreign particles can also be detected indirectly by opsonic receptors. The receptors for antibody and complement are the best described opsonic receptors.
The Archaeal Ancestry Of Actins
Bacteria and archaea encode well-characterized proteins homologous to actin such as MreB, FtsA, ParM, and MamK . MreB is essential for rod-shape maintenance and chromosome partitioning of bacteria, FtsA interacts with the tubulin-like FtsZ ring and is involved in cell division, whereas ParM is a plasmid-encoded protein required for segregation. The MreB and ParM proteins are structurally similar to actin and polymerize to form filaments . The MamK protein is essential for the spatial organization of magnetosomes and also forms filamentous structures .
The sequence similarity between actins and their closest bacterial homologs, MreB and FtsA, is extremely low despite the conservation of the main structural elements of the RNAseH/HSP70 fold and the amino acid residues that are essential for the ATPase activity . This limited sequence conservation parallels the case of tubulin that is weakly similar to the bacterial/archaeal homolog, FtsZ, and sharply contrasts the high level of conservation between eukaryotic and bacterial Hsp70 chaperones, proteins that possess the same core fold as actins. Considering the central importance of the cytoskeleton for the very existence of eukaryotic cells, deciphering the underlying causes of the dramatic sequence divergence between actin is central to the understanding of eukaryotic origins. At least, two distinct although not necessarily incompatible hypotheses can be considered.
Figure 1Figure 2
You May Like: Algebra Homework Help Free Online
Nursing Allied Health And Interprofessional Team Interventions
The management of resistant staphylococcal infections requires a dedicated interprofessional team, relying on the multidisciplinary skill sets of individuals in pharmacology, medicine, nursing, and infectious disease. The rate of iatrogenic disease spread must be contained through team efforts of proper handwashing and the use of personal protective equipment . Identifying appropriate treatment modalities that account for resistance mechanisms must be employed by physicians and pharmacologists, while patient monitoring under nursing team members is crucial to the patient and family members safety. Acute hospital care knowledge regarding MRSA management, infection source, and geographic variability of resistant strains is crucial to treatment outcomes.
Disruption Of Cell Signaling
Leishmania tropica
Some survival strategies often involve disrupting cytokines and other methods of cell signaling to prevent the phagocyte’s responding to invasion. The protozoan parasites Toxoplasma gondii, Trypanosoma cruzi, and Leishmania infect macrophages, and each has a unique way of taming them. Some species of Leishmania alter the infected macrophage’s signalling, repress the production of cytokines and microbicidal moleculesânitric oxide and reactive oxygen speciesâand compromise antigen presentation.
Read Also: What Is Scaffolding In Psychology
Reviewer’s Report : Pierre Pontarotti
The authors carried out a comparative analysis between the phagosome of eukaryote from highly divergent phyla by using data from proteomic analyses and literature searches.
To extend their analyses, they subsequently checked for the presence of these conserved eukaryote genes in archaea and bacteria.
They found that the orthologues of one of such a gene are present in subset of archaea. This gene is orthologous to eukaryotic actins gene and share unusual structural features with actins related proteins 2 and 3. The authors deduced that the presence of common structural features in Arp/23 and the archea actins, implies that the common ancestors between the archaea and eukaryotic actins were able to develop branch filaments.
They further found that the orthologue of the Rho family appeared to be of bacterial origin.
These two findings lead the authors to hypothesize than the ancestor of the eukaryotes was an archeon that had an actin-based-cytoskeleton allowing engulfment of one bacteria , horizontal transfer then occurred leading to eukaryogenesis.
I think that this hypothesis on the origin of the eukaryotic phyla is of great interest. However the authors’ conclusion is only based on two genes .
My first recommendation is to change the title as follows: “the possible origins of phagocytosis and eukaryogenesis” that should better describe the works of authors.
Other comments:
If the corresponding genes are found, then two explanations are possible
Receptors For Foreign Particles
2.1.1. Pattern-Recognition Receptors
Some receptors that directly bind PAMPs and seem to be phagocytic receptors include Dectin-1, mannose receptors, CD14, and scavenger receptor A . Dectin-1 binds to polysaccharides of some yeast cells . Mannose receptors bind mannan . CD14 binds to lipopolysaccharide-binding protein . SR-A can detect lipopolysaccharide on some gram-negative bacteria and on Neisseria meningitidis . Among these receptors, Dectin-1 has been clearly shown to be sufficient for activating phagocytosis. When it is expressed on heterologous cells that normally cannot perform phagocytosis, it gives the cells phagocytic capabilities . However, for other PAMP receptors the phagocytic potential is still a matter of debate. It may be that they induce phagocytosis indirectly by tethering the particle to the phagocyte surface, or by priming the phagocyte to ingest the particle via other receptors.
You May Like: How Does Physical Geography Affect Military Strategy And Planning
Ingestion Of The Microbe Or Cell By The Phagocyte
Following attachment, polymerization and then depolymerization of actin filaments send pseudopods out to engulf the microbe ) and place it in an endocytic vesicle called a phagosome ).
| html5 version of animation for iPad showing ingestion and phagosome formation. |
During this process, an electron pump brings protons into the phagosome. This lowers the pH within the phagosome to 3.5 – 4.0 so that when a lysosome fuses with the phagosome, the pH is correct for the acid hydrolases to effectively break down cellular proteins. The acidification also releases defensins, cathelicidin, and bacterial permeability inducing protein , peptides and enzymes that can kill microbes, from a matrix and enabling their activation.
| You Tube Movie illustrating a neutrophil phagocytosing MRSA |
Intracellular microbes, such as viruses and bacteria that invade host cells, can also be engulfed once they enter the cytosol of the cell by a process called autophagy. A membrane-bound compartment called an autophagosome grows around the microbe and the surrounding cytosol and subsequently delivers it to lysosomes for destruction ).
Some microorganisms are more resistant to phagocytic ingestion
a. Pathogenic Yersinia, such as the one that causes plague, contact phagocytes and, by means of a type III secretion system, deliver proteins which depolymerize the actin microfilaments needed for phagocytic engulfment into the phagocytes ). Another Yersinia protein degrades C3b and C5a.
Reviewer’s Report : Gaspar Jekely Max Planck Institute For Developmental Biology Tbingen
This interesting paper by Yutin et al. addresses one of the most important and hotly debated issues about the origin of eukaryotes, namely the origin of phagocytosis.
It is now clear that the last common ancestor of eukaryotes was able to perform phagocytosis, and possessed essentially all the characteristic eukaryotic organelles, including mitochondria. The debate is about whether phagotrophy or mitochondria came first. Since both evolved in the stem lineage leading to modern eukaryotes, the order of origins is problematic. Cell biological considerations rather favor a phagotrophic host for the mitochondrium, but there are also strong advocates for the alternative.
One way to test the two models would by a thorough phylogenetic analysis of the proteins involved in phagocytosis. If most of these proteins turned out to be of alpha-proteobacterial origin, this would favor the mitochondrium-first scenario. Conversely, if most proteins were similar to ones found in archaebacteria, the sister lineage to the eukaryotic host, the phagotrophy-first scenario would gain support.
Yutin et al. try to resolve the issue exactly this way. In my opinion, however, they fail to find a conclusive answer, or more precisely, the data fail to yield a clear support for either scenario. The solution they propose is somewhat arbitrary, at least if we want to conclude something strictly based on the data presented. I will explain why I think this is the case.
Read Also: What Is Real Quantum Physics
The Mechanism Of Phagocytosis
Owing to the extreme diversity in phagocyte and target particle types, phagocytosis has evolved into a highly complex phenomenon in higher organisms. Despite this complexity, the basic mechanisms of particle internalization and degradation remain conserved and is discussed below.
Schematic depicting the mechanisms behind phagocytosis
Phagocytosis is initiated by the binding of specialized receptors expressed on the phagocyte cell membranes to distinct molecular patterns on the surfaces of the target particles. Binding is followed by actin polymerization at the site of ingestion, which causes extensive deformation of the plasma membrane into pseudopodial extensions. The pseudopodia surrounds the particle completely so that the particle is engulfed in a cup-shaped structure, called the phagocytic cup. Once the particle has been internalized, actin filaments begin depolymerizing from the base of the phagocytic cup, causing the cup to close into a membrane-bound vacuole called the phagosome. Thereafter, the phagosome undergoes maturation, through a series of membrane fusion and fission events with the endosomal compartments, until it fuses with the lysosomes to form a highly microbicidal phagolysosome .
Activation Of The Internalization Process
When a particle is recognized by phagocytic receptors, various signaling pathways are activated to initiate phagocytosis. Reorganization of the actin cytoskeleton and changes in the membrane take place resulting in a depression of the membrane area touching the particle, the phagocytic cup. Then, pseudopods are formed around the particle until the membrane completely covers the particle to form a new phagosome inside the cell. The signaling mechanisms to activate phagocytosis are best-known for Fc receptors and for complement receptors . For other phagocytic receptors, signaling pathways are just beginning to be investigated.
Also Check: What Does Expression Mean In Math
Host Damage By Phagocytes
Macrophages and neutrophils, in particular, play a central role in the inflammatory process by releasing proteins and small-molecule inflammatory mediators that control infection but can damage host tissue. In general, phagocytes aim to destroy pathogens by engulfing them and subjecting them to a battery of toxic chemicals inside a phagolysosome. If a phagocyte fails to engulf its target, these toxic agents can be released into the environment . As these agents are also toxic to host cells, they can cause extensive damage to healthy cells and tissues.
When neutrophils release their granule contents in the kidney, the contents of the granule degrade the extracellular matrix of host cells and can cause damage to glomerular cells, affecting their ability to filter blood and causing changes in shape. In addition, phospholipase products intensify the damage. This release of substances promotes chemotaxis of more neutrophils to the site of infection, and glomerular cells can be damaged further by the adhesion molecules during the migration of neutrophils. The injury done to the glomerular cells can cause kidney failure.
Phagocytosis: Our Current Understanding Of A Universal Biological Process
- 1División de Estudios de Posgrado e Investigación, Facultad de Odontología, Universidad Nacional Autónoma de México, Mexico City, Mexico
- 2Departamento de Inmunología, Instituto de Investigaciones Biomédicas, Universidad Nacional Autónoma de México, Mexico City, Mexico
Phagocytosis is a cellular process for ingesting and eliminating particles larger than 0.5 m in diameter, including microorganisms, foreign substances, and apoptotic cells. Phagocytosis is found in many types of cells and it is, in consequence an essential process for tissue homeostasis. However, only specialized cells termed professional phagocytes accomplish phagocytosis with high efficiency. Macrophages, neutrophils, monocytes, dendritic cells, and osteoclasts are among these dedicated cells. These professional phagocytes express several phagocytic receptors that activate signaling pathways resulting in phagocytosis. The process of phagocytosis involves several phases: i) detection of the particle to be ingested, ii) activation of the internalization process, iii) formation of a specialized vacuole called phagosome, and iv) maturation of the phagosome to transform it into a phagolysosome. In this review, we present a general view of our current understanding on cells, phagocytic receptors and phases involved in phagocytosis.
Read Also: Geometry Unit 4 Practice Answers
The Core Set Of Proteins Involved In Phagocytosis Identified By Comparison Of The Proteomics Results
Table 1 Key receptor proteins involved in phagocytosisa
Obviously, the collections of phagosomal proteins identified by proteomic approaches include numerous false positives and false negatives. The false-positive group seems to consist, primarily, of some of the abundant cytosolic and organellar proteins that contaminate the isolated phagosomes. The false-negative group would include proteins that, although functionally important, only transiently associate with the phagosome, and therefore, escape detection by proteomics. Accordingly, we attempted to correct and extend the list of phagosomal proteins by including proteins that have not been consistently detected by proteomics but whose role in phagocytosis has been convincingly demonstrated in at least two organisms, and excluding proteins without concrete functional roles in phagocytosis.
Differences Between Pinocytosis And Phagocytosis
Pinocytosis and phagocytosis are similar processes, but they have some key differences. Phagocytosis is used to absorb materials much bigger than the particles that can be absorbed by pinocytosis, such as bacteria. Phagocytosis involves the ingestion of solid materials, while pinocytosis involves the ingestion of liquids and solute particles. During pinocytosis, the contents of the vesicles are emptied directly into the cell, but this doesnt happen in phagocytosis because the contents of the vesicles are too big. Lysosomes must combine with the vesicles to break down the contents. This process does not occur in pinocytosis.
Don’t Miss: What Is Imf In Chemistry
The Bacterial Roots Of Eukaryotic Small Gtpases
Figure 3
A Maximum Likelihood tree of the Ras superfamily of GTPases. The tree was constructed using 120 aligned positions . The tree is unrooted but shown in a pseudo-rooted form solely for convenience. Bacterial and archaeal clusters are shown in green and red, respectively. Support values are shown only for major internal branches. For the complete legend, see Additional File .
Icle Recognition: Phagocytosis Receptors
Phagocytes must recognize many different particles that could potentially be ingested, including all classes of pathogens and dying cells. Phagocytes investigate their surroundings for phagocytic particle targets by extending finger-like membrane projections called filopodia. Various receptors on the plasma membrane of phagocytic cells recognize these particles and then initiate the process of phagocytosis. A single phagocyte can express several receptor types, and they cooperate for the recognition and ingestion of the particle. Phagocytic receptors are classified as non-opsonic receptors or as opsonic receptors. Non-opsonic receptors can directly recognize pathogen associated molecular patterns and also activate the phagocytosis process . These receptors include C-type lectins, such as Dectin-1 , Dectin-2, DC-SIGN and Mincle and also scavenger receptors . Toll-like receptors can also detect molecular patterns on pathogens, but they are not phagocytic receptors . However, TLRs can cooperate with phagocytic receptors to make the phagocytic process more effective .
Table 4. Non-opsonic receptors.
| IgG | Nimmerjahn and Ravetch |
Don’t Miss: Kuta Software Infinite Geometry Angle Pair Relationship
Survival Inside The Phagocyte
Bacteria have developed ways to survive inside phagocytes, where they continue to evade the immune system. To get safely inside the phagocyte they express proteins called invasins. When inside the cell they remain in the cytoplasm and avoid toxic chemicals contained in the phagolysosomes. Some bacteria prevent the fusion of a phagosome and lysosome, to form the phagolysosome. Other pathogens, such as Leishmania, create a highly modified vacuole inside the phagocyte, which helps them persist and replicate. Some bacteria are capable of living inside of the phagolysosome. Staphylococcus aureus, for example, produces the enzymes catalase and superoxide dismutase, which break down chemicalsâsuch as hydrogen peroxideâproduced by phagocytes to kill bacteria. Bacteria may escape from the phagosome before the formation of the phagolysosome: Listeria monocytogenes can make a hole in the phagosome wall using enzymes called listeriolysin O and phospholipase C.
Examples Of Phagocytosis In A Sentence
phagocytosisForbesphagocytosisWiredphagocytosisScientific American
These example sentences are selected automatically from various online news sources to reflect current usage of the word ‘phagocytosis.’ Views expressed in the examples do not represent the opinion of Merriam-Webster or its editors. Send us feedback.
Don’t Miss: Do You Need Physics For Occupational Therapy
Definition: What Is Phaocytosis
Essentially, phagocytosis may be described as a form of endocytosis through which a cell engulfs particulate matter/solid particles/cells. For different types of cells, phagocytosis plays a number of different roles ranging from food ingestion to the destruction of given cells and particulate matter.
Some of the cells that use phagocytosis include:
- Some vascular endothelial cells
* Generally, phagocytes are divided into two major groups. These include “professional phagocytes” and “non-professional phagocytes” .
Living cells take in different types of material across their cell membrane. For the most part, a majority of these material/molecules such as ions, fluids, and oxygen among others easily pass through the membrane through such mechanisms as ion pumps and osmosis among others.
Some of the matter, e.g. particles like viruses, may prove too large to pass through the membrane through such mechanisms. For this reason, the cell has to engulf such matter/objects into the cell.
This process involves the invagination of the cell membrane in question which allows the cell to take in the object/particle. Depending on the cell and the mechanism used to engulf such material/objects, endocytosis is divided into phagocytosis, pinocytosis and another process known as receptor-mediated endocytosis.
Step 1: Activation/ actuation
Step 2: Binding