Resonance Structures Of Benzene
Benzene is a very important aromatic hydrocarbon in organic chemistry. It has the chemical formula C6H6. The molecules of benzene have a cyclic structure consisting of alternating single and double bonds between adjacent carbon atoms. Each carbon atom is also bonded to one hydrogen atom. The two possible resonance structures of benzene are illustrated below.
The benzene molecule is stabilized by resonance, the pi electrons are delocalized around the ring structure. This delocalization causes each carbon-carbon bond to have a bond order of 1.5, implying that they are stronger than regular C-C sigma bonds. In the resonance hybrid of benzene, the delocalization of pi electrons is described with the help of a circle inside the hexagonal ring.
In benzene, Kekules first suggested two cyclohexatriene Kekule structures that have been taken together, they constitute the general structure as contributing structures. The hexagon replaces three double bonds in the hybrid structure on the right and represents six electrons in a collection of three molecular orbitals with a nodal plane in the molecule plane.
Thus, the resonance structures of some molecules and polyatomic ions are discussed briefly in this article. Learn more about this concept and other related concepts such as hyperconjugation, resonance effect, and electron dot formula.
Resonance Effect Or Mesomeric Effect In Chemistry
The withdrawal effect or releasing effect of electrons attributed to a particular substituent through the delocalization of or pi-electrons that can be seen by drawing various canonical structures is called a resonance effect or mesomeric effect. M or R symbols are used to represent the resonance effect.
The above figure shows the different resonance structures of different compounds with resonance effects.
The concept of resonance effect tells about the polarity induced in a molecule by the reaction between a lone pair of electron and a pi bond. It also occurs by the interaction of 2 pi bonds in the adjacent atoms. Resonance in simple is the molecules with multiple Lewis structures. Resonance in chemistry helps in understanding the stability of a compound along with the energy states.
Definition The resonance effect can be defined as a chemical phenomenon that is observed in the characteristic compounds having double bonds in the organic compounds. The organic compounds contain these double bonds in the structures and usually have the overlapping of the p-orbitals on the two adjacent sides of carbon atoms.
Resonance Hybrid And Movement Of Electrons
I know that I have just told you that curved arrows show the movement of electrons but I also need to tell you something that goes against this.
Even though we use curved arrows and move the electrons around in resonance structures, you need to know that the electrons do not actually move in the sense of jumping from one atom to another as we show them in resonance structures.
In reality, the electrons are spread among the atoms and none of the resonance forms is the correct representation of the molecule.
The accurate representation of the molecule is given by the resonance hybrid.
Having the resonance forms in brackets is to indicate that they represent one entity, which is the resonance hybrid where the charge are spread over the two atoms.
You may wonder now why do we then draw resonance structures and use curved arrows? And the answer to this is that some properties and reactions of molecules are better explained by the individual resonance structures and we use them with curved arrows to keep track of electrons and explain these properties.
Think about a hybrid fruit nectarine. It is a mix of a peach and a plum and to explain its color, texture, and the taste, we refer to the individual fruits.
This, however, does not mean that the nectarine exists as a peach for some time and then turns into a plum. None of them is a correct representation of the nectarine just like none of the resonance structures is the correct representation of the given molecule.
Also Check: What Does Compass Rose Mean In Geography
Resonance And Peptide Bonds
What is the hybridization state of the nitrogen atom in an amide? At first glance, it would seem logical to say that it is sp3-hybridized, because, like the nitrogen in an amine, the Lewis structure shows three single bonds and a lone pair. The picture looks quite different, though, if we consider another resonance contributor in which the nitrogen has a double bond to the carbonyl carbon: in this case, we would have to say that applicable hybridization is sp2, and the bonding geometry trigonal planar rather than tetrahedral.
In fact, the latter picture is more accurate: the lone pair of electrons on an amide nitrogen are not localized in an sp3 orbital, rather, they are delocalized as part of a conjugated pi system, and the bonding geometry around the nitrogen is trigonal planar as expected for sp2 hybridization. This is a good illustration of an important point: conjugation and the corresponding delocalization of electron density is stabilizing, thus if conjugation can occur, it probably will.
One of the most important examples of amide groups in nature is the âpeptide bondâ that links amino acids to form polypeptides and proteins.
The barrier to rotation in peptide bonds is an integral part of protein structure, introducing more rigidity to the protein’s backbone. If there were no barrier to rotation in a peptide bond, proteins would be much more ‘floppy’ and three dimensional folding would be very different.
Exercise 2.20
Exercise 2.21
Exercise 2.22
A word of advice
Are Resonance Structures Important In Organic Chemistry
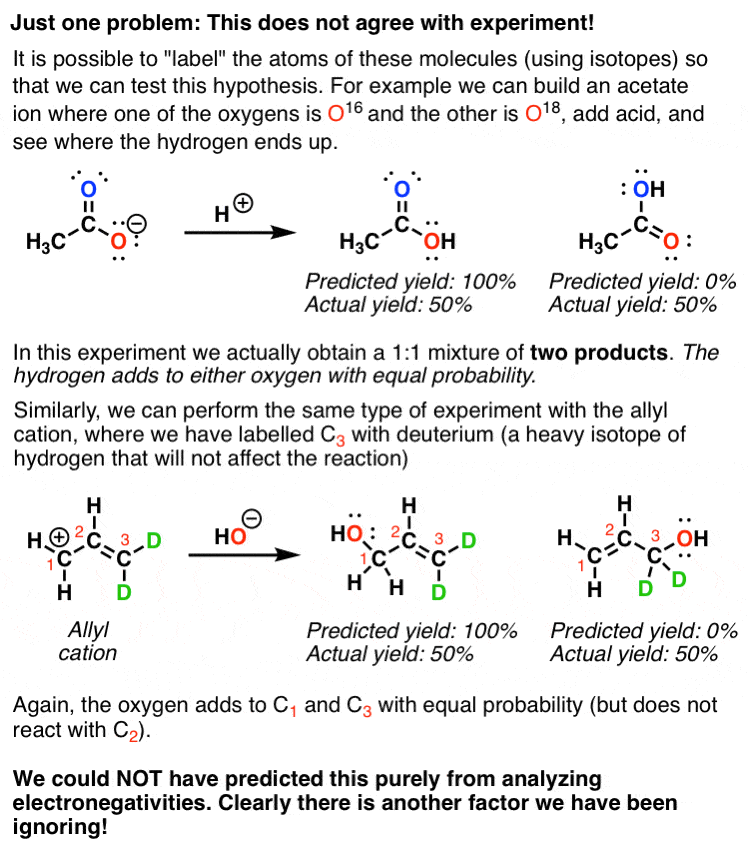
Resonance Structures. Resonance Structures: How to get them! Resonance is probably the single most important concept in organic chemistry since if resonance is present, then it typically controls the outcome. One of the key applications is understanding substituent effects, particularly in aromatic systems.
Recommended Reading: Alphabetical List Of Geometry Terms
Resonance Structures Organic Chemistry Tutorial Video
September 26, 2016 By Leah4sci
Orgo Basics Video Series: Video 6 Resonance structures are alternative Lewis structures for a molecule or an ion. To draw the resonance hybrid or structures, we are moving electrons between adjacent atoms. Overwhelmed?
In the video below, you will see an explanation and illustration of resonance structures and resonance hybrids step-by-step. This will help you visualize what is going on behind arrows.
Factors That Influence These Effects:
Inductive Effect: The degree of inductive impact is affected by the electronegativity values of atoms.
Resonance Effect: The resonance effect is influenced by the number of double bonds and their order.
The induced electrical charges in atoms of a molecule generate the inductive effect. The difference in the electronegativity values of atoms causes charge induction. Atoms with a high electronegativity have a tendency to attract bond electrons. The resonance effect, on the other hand, is distinct from the inductive effect. The resonance effect of a molecule occurs when the molecule has double bonds. The inductive effect describes the transmission of electrical charges between atoms in a molecule, whereas the resonance effect describes the transmission of electron pairs between atoms in a molecule.
Also Check: What Is Protein In Chemistry
Condition For Resonance In Chemistry
Resonance is popular in Organic Chemistry. When we hear the word resonance, the questions that primarily come to mind are – ‘What is Resonance?’, ‘What is the condition for Resonance?’.
Table of Content
In the case of certain molecules, a single Lewis structure cannot explain all the molecules properties. Under the framework of VBT, resonance is an extrapolated idea that a Lewis structure can describe the bonding in a chemical species. For many chemical species consisting of atoms obeying the octet rules, possibly bearing the formal charges and connected by bonds of positive integer order, it is not sufficient for describing all properties. In this situation, several contributing structures are helped to understand the properties in this case, resonance is considered. As we have answered the first question, we will discuss the condition for resonance in chemistry.*-
What Is Resonance Effect
We cannot predict the properties of many organic compounds with the help of single Lewis dot structure. For example, lets consider the case of benzene. Going by the Lewis dot method, we would end up predicting Benzene to have three C-C bonds and three C=C bonds. But, the actual property deviates from this prediction.
Thus, we define resonance structures for defining properties of these compounds. The resonance structures are actually hypothetical. This is because they do not represent any real molecule individually. They contribute to the actual structure in proportionately according to their stability.
The energy of actual structure of the molecule is lower than that of any of the canonical structures.The resonance energy increases with the number of important contributing structures. The number of unpaired electrons is the same in the resonance structures and so also are the positions of nuclei.
Read Also: What Is Another Word For Average In Math
Quantum Mechanical Aspect Of Resonance
Resonance has deep importance in the mathematical formalism of Valence Bond Theory. Quantum Mechanics requires the wave function of a molecule to obey its observed symmetry. If a single contributing structure does not match this, resonance is invoked. For example, Benzene VBT begins with 2 Kekule structures that do not individually possess the six pole symmetry of the whole molecule. The theory constructs the actual wave function as a linear superposition of the wave functions representing the two structures. As both Kekule structures have equal energy, they are equal contributors to the overall structure. The superposition is an equally-weighted average 1:1 linear combination of the two in the case of Benzene. The symmetric combination gives the ground state, while the antisymmetric combination gives the first excited state. The superposition or resonance is written with undetermined coefficients, which are then variationally optimised to find the lowest possible energy for the given set of basis wavefunctions. When more contributing structures are included, the molecular wave function becomes more accurate, and more excited states can be derived from different combinations of contributing structures.
Resonance Structures Of No3 Ion
Nitrogen is the central atom in a nitrate ion. It is singly bonded to two oxygen atoms and doubly bonded to one oxygen atom. The oxygen atoms that are singly bonded to the nitrogen hold a charge of -1 . The central nitrogen atom has a charge of +1 and the overall charge on the nitrate ion is -1. The three possible resonance structures of NO3 are illustrated below.
If a resonance hybrid of this polyatomic ion is drawn from the set of Lewis structures provided above, the partial charge on each oxygen atom will be equal to -. The net charge on the central atom remains +1. This resonance hybrid is illustrated below.
You May Like: How Did Geography Of Rome Affect Its Expansion
Resonance Structures In Organic Chemistry
Resonance stabilization effect , as briefly mentioned in Section 1.3, is one of the fundamental concepts of Organic Chemistry and has broad applications. The discussion of resonance effect heavily relies on the understanding of resonance structures. Here we will focus on how to draw resonance structures for organic chemistry species, and how to compare the relative stabilities between the structures.
According to resonance effect, the greater the number of resonance contributors, the greater the resonance stabilization effect, and the more stable the species is. Therefore, to predict whether the resonance effect applies or not, we usually need to construct ânewâ resonance structures based on the âoriginalâ one that is available. There are some very important rules we need to follow for such purposes.
Guidelines for Drawing Resonance Structures:
arrow pushing
Examples: Draw another resonance structure based on the given one.
Resonance Structure And Dominance
In some examples of resonance, the multiple resonance structures contribute equally to the overall hybrid structure. For example, earlier we looked at ozone. It can be described using two resonance structures. The overall hybrid structure is a perfect average of the two. However, in some cases, one structure has more influence than the others. We say that this structure is dominant. The dominant structure is determined using formal charges.
Formal charges are charges assigned to atoms, assuming that all the bonded electrons are split evenly between the two bonded atoms.
We have a whole article dedicated to formal charges, where you can find out how to calculate them for all sorts of molecules. Head over to “Formal Charges” for more.
In general, we assume that the Lewis structure with formal charges closest to zero is the dominant structure. If two resonance structures both have equivalent formal charges, we assume that the Lewis structure with the negative formal charge on the more electronegative atom is the dominant structure.
Take a look at the three possible resonance structures of carbon dioxide, shown below. In two of the structures, shown in the middle and on the right, one of the oxygen atoms has a formal charge of +1 and the other has a formal charge of -1. In the other resonance structure, shown on the left, all atoms have a formal charge of +0. This is therefore the dominant structure.
Dominant structure in resonance. StudySmarter Originals
Also Check: How To Get Answers On Delta Math
What Can You Change When Drawing Resonance Structures
Rules to draw resonance structures Structures are changed only with their electrons configuration. Number of total electron pairs should be same in every structure. Total electron pairs can be simplified as bonds and lone pairs. Otherwise we can say, number of electron pairs = bonds + bonds + lone pairs.
Rules For Drawing Resonance Structures
Two must-follow rules when drawing resonance structures:
1) Do not exceed the octet on 2nd-row elements.
2) Do not break single bonds
Rule 1. The second-row elements can only handle up to eight electrons because of their orbitals. And this means, you should never place more than eight electrons on those, i.e. you must follow the octet rule.
They may have less than eight electrons, but never more. Read this post to refresh standard valences and formal charges in organic chemistry.
Rule 2:Do not break single bonds. The basis of this rule is that atoms must have the same placement in resonance structures otherwise they are not resonance structures but rather different molecules.
You can only move electrons in writing resonance structures if it is not changing the way the atoms are connected. So, one way of drawing a resonance structure above would be starting the arrow from the lone pair and then break the bond:
One good pattern to remember is that resonance structures involve a bond, one way or the other. It is either making a bond or breaking a bond or both.
Therefore, whenever asked to draw a resonance structure, look for a bond. You cant have resonance structures with having a bond involved.
If there is no bond, then it would have to be formed in the new resonance structure.
Read Also: What Kind Of Science Is Chemistry
Its Time For Resonance In Organic Chemistry
Resonance in organic chemistry is one of the most fundamental and useful concepts you will learn in this class. Once most students hear this tip, it makes perfect sense to them, but it isnt one that you might think of on your own. Take a look at the structure below, and ask yourself: are the two N-O bonds in this molecule the same length?
Since freshman chemistry, we have been told that double bonds between two atoms are shorter than a single bond between the same two atoms. Hence, the N-O double bond should be shorter than the N-O single bond. Spoiler: it is not. But before we get into that, lets look at some resonance forms of the nitro group at the end of this hydrocarbon:
Here, we can more clearly see that the nitro group is interconverting between the three resonance structures shown above. Structure 3, where the charge is spread evenly between the two oxygens is a valid structure and shows that the bond two oxygen atoms in the molecule are equivalent and have the same bond length . This is shown here using the dashed bond, which you can think of as half of a bond for lack of a better term.
We care even more about this principle when it can be applied to more complex organic molecules where it is not obvious that the bonds are equivalent. For example, the cyclopentadiene anion:
Take Home Message: If you see symmetry or aromaticity, think equivalent bond lengths
What Is The Condition For Resonance
Ans. In chemistry , resonance is a path of understanding bonding character in certain molecules or ions by combining several contributing structures. Resonance occurs because of the overlap of orbitals. The planarity of a molecule plays a vital role in resonance.
Ans. Benzene is such a molecule that has resonating structures. 1,3 Butadiene is also a molecule with resonating structures .
Ans. The energy of the structures, the position of nuclei of structures, and the number of non-bonding pairs of electrons are similar between the different resonating structures.
Ans. Resonance forms are hybrid, and hence it is not correct to say that they are in equilibrium. Equilibrium is altogether a different concept.
Ans. The position of two pi bonds in conjugation is the main condition for resonance. The planarity of the molecule also plays a very important role in resonance these are the key points that answer the question What is the condition for Resonance?
Read Also: Who You Spend Time With Is Who You Become Psychology
A Second Application Of Resonance: Pi Acceptors
When double bonds are connected to a polarized bond, the molecule will have a significant resonance form where there is positive charge on the adjacent carbon. This phenomenon is known as pi-accepting behavior, and these groups are known as pi acceptors.
For now, that does it for a summary of the important themes in resonance. Next stop will be to apply these principles to chemical reactivity.