Section : Units Of Measurement
International System of Units and the Metric System
The International System of Units, abbreviated SI from the French Système International Dunités, is the main system of measurement units used in science. Since the 1960s, the International System of Units has been internationally agreed upon as the standard metric system. The SI base units are based on physical standards. The definitions of the SI base units have been and continue to be modified and new base units added as advancements in science are made. Each SI base unit except the kilogram is described by stable properties of the universe.
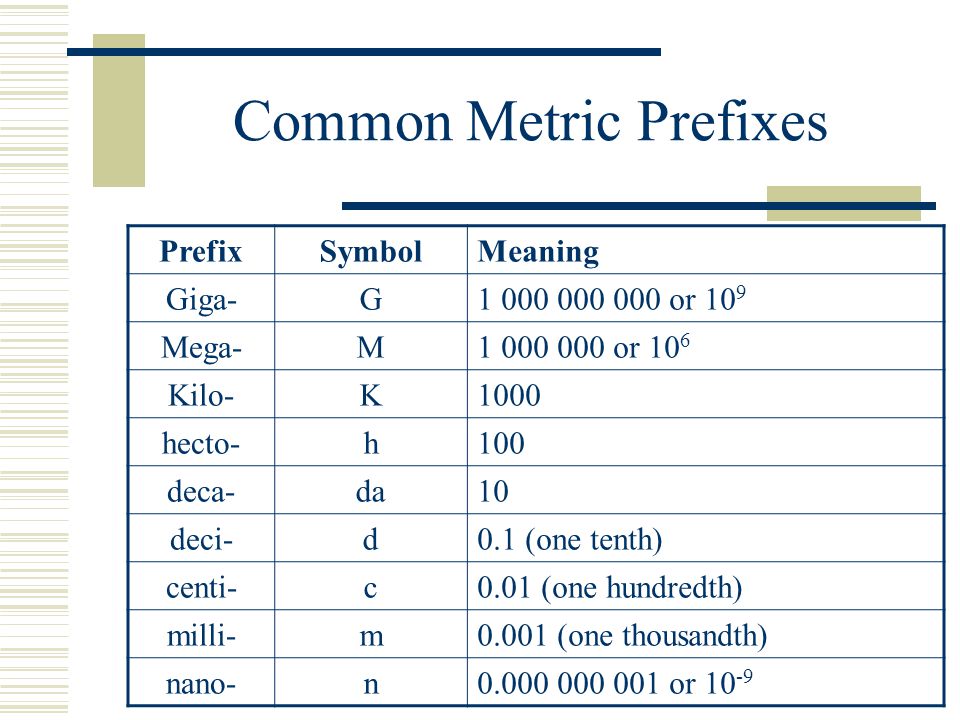
There are seven base units, which are listed in Table 1.2. Chemistry primarily uses five of the base units: the mole for amount, the kilogram for mass, the meter for length, the second for time, and the kelvin for temperature. The degree Celsius is also commonly used for temperature. The numerical relationship between kelvins and degrees Celsius is as follows
Sample Molecular Mass Calculation
Molecular mass may be calculated by taking the atomic mass of each element present and multiplying it by the number of atoms of that element in the molecular formula. Then, the number of atoms of each element is added together.
For example. to find the molecular mass of methane, CH4, the first step is to look up the atomic masses of carbon C and hydrogen H using a periodic table:
carbon atomic mass = 12.011hydrogen atomic mass = 1.00794
Because there is no subscript following the C, you know there is only one carbon atom present in methane. The subscript 4 following the H means there are four atoms of hydrogen in the compound. So, adding up the atomic masses, you get:
methane molecular mass = sum of carbon atomic masses + sum of hydrogen atomic masses
methane molecular mass = 12.011 +
methane atomic mass = 16.043
This value may be reported as a decimal number or as 16.043 Da or 16.043 amu.
Note the number of significant digits in the final value. The correct answer uses the smallest number of significant digits in the atomic masses, which in this case is the number in the atomic mass of carbon.
The molecular mass of C2H6 is approximately 30 or . Therefore the molecule is about 2.5 times as heavy as the 12C atom or about the same mass as the NO atom with a molecular mass of 30 or .
Section : How Scientists Study Chemistry
The Scientific Method
How do scientists work? Generally, they follow a process called the scientific method. The scientific method is an organized procedure for learning answers to questions. To find the answer to a question , a scientist goes through the following steps, which are also illustrated in Figure 1.7.
Figure 1.7 The General Steps of the Scientific Method. The steps may not be as clear-cut in real life as described here, but most scientific work follows this general outline.
Propose a hypothesis. A scientist generates a testable idea, or hypothesis, to try to answer a question or explain how the natural universe works. Some people use the word theory in place of hypothesis, but the word hypothesis is the proper word in science. For scientific applications, the word theory is a general statement that describes a large set of observations and data. A theory represents the highest level of scientific understanding, and is built from a wide array of factual knowledge or data.
Test the hypothesis. A scientist evaluates the hypothesis by devising and carrying out experiments to test it. If the hypothesis passes the test, it may be a proper answer to the question. If the hypothesis does not pass the test, it may not be a good answer.
Not all scientific investigations are simple enough to be separated into these three discrete steps. But these steps represent the general method by which scientists learn about our natural universe.
Recommended Reading: Vo In Physics
What Does Mm Stand For Chemistry
We compiled queries of the MM abbreviation in Chemistry in search engines. The most frequently asked MM acronym questions for Chemistry were selected and included on the site.
We thought you asked a similar MM question to the search engine to find the meaning of the MM full form in Chemistry, and we are sure that the following Chemistry MM query list will catch your attention.
Calculating Volume Of Solution
Calculate the volume of a 0.80 mol L-1 potassium bromide solution containing 1.60 moles of potassium bromide.
V = volume of solution in litres = ? L
Extract the data from the question: n = moles of solute = 1.60 mol c = molarity of solution = 0.80 mol L-1
Write the equation:
V = 1.60 ÷ 0.80 = 2.00 L
Can you apply this?
You May Like: My.hrw Answers
Nature Of The Particles
The mole is essentially a count of particles. Usually the particles counted are chemically identical entities, individually distinct. For example, a solution may contain a certain number of dissolved molecules that are more or less independent of each other. However, in a solid the constituent particles are fixed and bound in a lattice arrangement, yet they may be separable without losing their chemical identity. Thus the solid is composed of a certain number of moles of such particles. In yet other cases, such as diamond, where the entire crystal is essentially a single molecule, the mole is still used to express the number of atoms bound together, rather than a count of multiple molecules. Thus, common chemical conventions apply to the definition of the constituent particles of a substance, in other cases exact definitions may be specified.The mass of 1 mole of a substance is equal to its relative atomic or molecular mass in grams.
Problems Calculating Molecular Mass
While it’s possible to calculate molecular mass for small molecules, it’s problematic for polymers and macromolecules because they are so large and may not have a uniform formula throughout their volume. For proteins and polymers, experimental methods may be used to obtain an average molecular mass. Techniques used for this purpose include crystallography, static light scattering, and viscosity measurements.
Read Also: Glencoe Geometry Worksheets
Why We Use Moles
Why don’t we simply stick with units like grams ? The answer is that moles give us a consistent method to convert between atoms/molecules and grams. It’s simply a convenient unit to use when performing calculations. You may not find it too convenient when you are first learning how to use it, but once you become familiar with it, a mole will be as normal a unit as, say, a dozen or a byte.
How To Find Molarity
As long as you have a way of measuring the mass of a solute, you can calculate its molarity by measuring the volume of the solution. Be careful here, because molarity is always expressed as moles/liter, so if you measure volume in any other units, you have to convert to liters. Here are some conversion factors that you’ll find useful:
1 liter = 0.001 cubic meters = 1,000 milliliters = 0.264 U.S. gallons = 33.81 fluid ounces.
Also Check: How Did The Geography Of Greece Affect Its Development
What Does The 1 Superscript Mean In Units
For example, the rate of a chemical reaction can be expressed in $\mathrm/\mathrm^/\mathrm^$. Why is it 1 and not, say, 2? Does it change the meaning if the minus is removed and we simply express the rate in $\mathrm/\mathrm/\mathrm$?
- 15$\begingroup$The answers below are correct but none seem to mention that in math $x^$ equals $\dfrac$ for some variable $x$. The same thing applies here.$\endgroup$ Calvin’s HobbiesOct 21 ’14 at 21:51
- 5$\begingroup$Please note that a solidus shall not be followed by a multiplication sign or a division sign on the same line unless parentheses are inserted to avoid any ambiguity. Besides, the unit symbol for second is s .$\endgroup$ user7951
The -1 means “per” unit. So your first example mol/L-1/s-1 is not correct – it would actually be written as mol L-1 s-1, OR mol/. It is also sometimes written as mol/L/s, but the double division is ambiguous and should be avoided unless parentheses are used.
If it were mol L-1 s-2, this would mean moles per litre per second per second.
This is really just a question of notation, and is not chemistry-specific at all. Yes, all the minus/plus signs and the value of numbers are important. Good examples of units can include:
- area, measured in m2, or metres squared
- volume, measured in m3, or metres cubed
- pressure, measured in N m-2, or Newtons per metre squared
- velocity, measured in m s-1, or metres per second
- acceleration, measured in m s-2, or metres per second per second
What M And M Mean In Chemistry
Both m and M are units of the concentration of a chemical solution. The lowercase m indicates molality, which is calculated using moles of solute per kilograms of solvent. A solution using these units is called a molal solution . Uppercase M is molarity, which is moles of solute per liter of solution . A solution using this unit is termed a molar solution .
Molality = moles solute / kilograms solventThe units of molality are mol/kg.
Molarity = moles solute / liters solutionThe units of molarity are mol/L.
You May Like: John Thomas Child Of Rage
Redefinition Of Si Base Units
In 2011, the 24th meeting of the General Conference on Weights and Measures agreed to a plan for a possible revision of the SI base unit definitions at an undetermined date.
On 16 November 2018, after a meeting of scientists from more than 60 countries at the CGPM in Versailles, France, all SI base units were defined in terms of physical constants. This meant that each SI unit, including the mole, would not be defined in terms of any physical objects but rather they would be defined by constants that are, in their nature, exact.
Such changes officially came into effect on 20 May 2019. Following such changes, “one mole” of a substance was redefined as containing “exactly 6.02214076×1023 elementary entities” of that substance.
Section : Chemistry And Matter
What is Chemistry?
Everything around us is made up of chemicals. From the color that makes a rose so red to the gasoline that fills our cars and the silicon chips that power our computers and cell phonesChemistry is everywhere! Understanding how chemical molecules form and interact to create complex structures enables us to harness the power of chemistry and use it, just like a toolbox, to create many of the modern advances that we see today. This includes advances in medicine, communication, transportation, building infrastructure, food science and agriculture, and nearly every other technical field that you can imagine.
Chemistry is one branch of science. Science is the process by which we learn about the natural universe by observing, testing, and then generating models that explain our observations. is the process by which we learn about the natural universe by observing, testing, and then generating models that explain our observations. Because the physical universe is so vast, there are many different branches of science . Thus, chemistry is the study of matter, biology is the study of living things, and geology is the study of rocks and the earth. Mathematics is the language of science, and we will use it to communicate some of the ideas of chemistry.
Figure 1.1: The Relationships Between Some of the Major Branches of Science.Chemistry lies more or less in the middle, which emphasizes its importance to many branches of science.
Physical vs. Chemical Properties
You May Like: Colorful Overnight Geometry Dash
Popularity Rank By Frequency Of Use
How popular is MM among other acronyms?
- Search Molar Mass on Google
Discuss this MM abbreviation with the community:
Report Comment
We’re doing our best to make sure our content is useful, accurate and safe.If by any chance you spot an inappropriate comment while navigating through our website please use this form to let us know, and we’ll take care of it shortly.
When M And M Are Almost The Same
If your solvent is water at room temperature, m and M can be roughly the same, so if an exact concentration doesn’t matter, you can use either solution. The values are closest to each other when the amount of solute is small because molality is for kilograms of solvent, while molarity takes into account the volume of the entire solution. So, if the solute takes up a lot of volume in a solution, m and M won’t be as comparable.
This brings up a common mistake people make when preparing molar solutions. It’s important to dilute a molar solution to the correct volume rather than add a volume of solvent. For example, if you’re making 1 liter of a 1 M NaCl solution, you would first measure one mole of salt, add it to a beaker or volumetric flask, and then dilute the salt with water to reach the 1-liter mark. It is incorrect to mix one mole of salt and one liter of water.
Molality and molarity are not interchangeable at high solute concentrations, in situations where the temperature changes, or when the solvent is not water.
Read Also: Afda Mean Median Mode Range Practice
Section : Making Measurements In The Lab
Precision vs. Accuracy
It is important to note the different terminology we use when talking in science. One such set of terminology is precision and accuracy. Although precision and accuracy are often used interchangeably in the non-scientific community, the difference between the terms is extremely important to realize. Precision tells you how close two measurements are to one another, while accuracy tells you how close a measurement is to the known value. A measurement can be precise while not being accurate, or accurate but not precise the two terms are NOT related. A good analogy can be found in a game of darts . A player who always hits the same spot just to the left of the dart board would be precise but not very accurate. However, a dart player who is all over the board but hits the center of the board on average would be accurate but not precise. A good darts player, just like a good scientist, wants to be both precise and accurate.
Figure 1.11: Difference Between Accuracy and Precision. A game of darts can be used to show the difference between accuracy and precision.
Adapted from: https://upload.wikimedia.org/wikipedia/commons/thumb/5/5d/Reliability_and_validity.svg/717px-Reliability_and_validity.svg.png
Significant Figures
Figure 1.12: Measuring an Object to the Correct Number of Significant Figures.How many digits should be shown in this measurement?
Exact Numbers
Calculating Complicated Problems:
Conversions and the Importance of Units
Conversion Factors
Calculating Moles Of Solute
Calculate the moles of copper sulfate in 250.00 mL of 0.020 mol L-1 copper sulfate solution.
n = moles of solute = ? mol
Extract the data from the question: c = concentration of solution = 0.020 mol L-1 V = volume of solution = 250.00 mL = 250.00 ÷ 1000 = 250.00 x 10-3 L = 0.25 L
Write the equation:
n = 0.020 × 0.25 = 0.0050 mol
Don’t Miss: Which Is Harder Physics Or Chemistry
What Does The M Mean In Chemistry
. In this way, what does italicized m mean in chemistry?
Both m and M are units of the concentration of a chemical solution. The lowercase m indicates molality, which is calculated using moles of solute per kilograms of solvent. Uppercase M is molarity, which is moles of solute per liter of solution .
One may also ask, is M the same as mol L? Molar concentration. In chemistry, the most commonly used unit for molarity is the number of moles per litre, having the unit symbol mol/L. A solution with a concentration of 1 mol/L is said to be 1 molar, commonly designated as 1 M.
Hereof, what does 1.0 m mean in chemistry?
Molar can also apply to the concentration of a solute in water: a 1.0 Molar solution has 1.0 moles of solute made up to 1.0 liters of solution. This would be labeled 0.1 M HCl or 0.1 M Hydrochloric Acid. A mole is 6.02 x 10^23 atoms or molecules of a given substance.
What does 0.05m mean?
This means that a 1 M solution is 40g in 1 litre of water. A 0.05 M solution will be 40*0.05 g in a litre. This is 2g in a litre.
Calculating Molality Given Mass
If we mass 5.36 g of KCl and dissolve this solid in 56 mL of water, what is the molality of the solution? Remember that molality is moles of solute/kg per solvent. KCl is our solute, while water is our solvent. We will first need to calculate the amount of moles present in 5.36 g of KCl:
\text = 5.36 \text \times = 0.0719 \text
We also need to convert the the 56.0 mL of water to its equivalent mass in grams by using the known density of water :
56.0\ \text \times = 56.0\ \text
56.0 g of water is equivalent to 0.056 kg of water. With this information, we can divide the moles of solute by the kg of solvent to find the molality of the solution:
\text = = = 1.3\ \text
The molality of our KCl and water solution is 1.3 m. Since the solution is very dilute, the molality is almost identical to the molarity of the solution, which is 1.3 M.
Recommended Reading: What Does Span Mean Linear Algebra
What Does M M Mean In Chemistry
Accordingly, what mM means?
MMMM
One may also ask, how do you calculate equivalents?
calculateequivalents
How do I convert Kmol to Mol?
Quick conversion chart of kmol to mol
What MM stand for?
mmmillimeters