Growth Factor Reservoir And Release
The ECM might serve as a reservoir for factors that aid in the sustenance of CSCs. Embryonic stem cells have been shown to utilize matrix metalloproteases 1 to release ciliary neurotropic factor from an ESC-derived matrix, which enhances ESC self-renewal though JAK/STAT3 signaling , a pathway that has also been implicated in promoting self-renewal of breast CSCs . Hematopoietic stem cells also upregulate MMP-9 to release soluble kit-ligand, also known as stem cell factor , which promotes survival signaling and chemoresistance in many types of cancers . CSCs are thought to remodel their matrices more significantly than their non-cancer stem cell counterparts as CSCs upregulate expression of different MMPs. This may enable them to effectively degrade and remodel ECM matrices to release growth factors and cytokines to promote their survival.
Fibrous Proteins Collagen And Elastin
The collagens constitute what is now known to be a highly specialized family of glycoproteins. At present, 20 genetically distinct types encoded by at least 30 genes have been described. All collagens contain a domain with a triple helical conformation and are integral components of the extracellular matrix.
In triple helical domains three polypeptide chains, each with a left-handed conformation, wind round each other to form a right-handed superhelix. Every third residue is glycine and of the repeating triplet that makes up the chain, approximately 20% of X and Y are imino acids, proline and hydroxyproline. This triple helical domain varies in length for the different collagen types and can either be continuous or interrupted with nonhelical domains.
The molecular configuration and tissue distribution of some of the collagens are shown in Table 1. Attempts have been made to classify collagens, both with regard to their gene organization and supramolecular structure, and while some of the more recently described collagens cannot be categorized in this way, it does provide a useful guide as to their role in the ECM.
Table 1. The collagen family
| Collagen types | |
|---|---|
| FACIT? | Unknown |
Andre Zeug, … Evgeni Ponimaskin, in, 2014
Mmp Adamts And Adam Metalloproteinases
Matrix metalloproteinases and the related ADAMs and ADAMTSs are subfamilies of proteolytic enzymes belonging to the metzincin clan. They have important roles in ECM homeostasis and regulation of autocrine and paracrine signaling. MMPs and ADAMTSs are secreted enzymes, although some MMPs and several ADAMTSs are localized on the cell surface or in the pericellular environment ]. ADAMs, on the other hand, are TM metalloproteinases that act primarily on membrane-localized substrates.
Matrix metalloproteinases, ADAMTSs, and ADAMs are modular enzymes, containing a conserved catalytic domain and various ancillary domains that regulate substrate selectivity and enzyme localization. As the name suggests, the proteolytic activity of metzincins is dependent on an active site Zn2+ ion, which is coordinated by 3 histidine residues in the conserved HEXXHXXGXX consensus motif ]. The glutamic acid residue in this motif activates a Zn2+-bound H2O molecule to generate the nucleophile that cleaves substrate peptide bonds. Metzincins also have a conserved methionine residue downstream of the catalytic motif, forming the Met-turn that dictates the architecture of the active site.
Recommended Reading: What Does Alpha Mean In Chemistry
The Role Of The Extracellular Matrix In Cancer Stemness
- Department of Regenerative and Cancer Cell Biology, Albany Medical College, Albany, NY, United States
As our understanding of cancer cell biology progresses, it has become clear that tumors are a heterogenous mixture of different cell populations, some of which contain so called cancer stem cells . Hallmarks of CSCs include self-renewing capability, tumor-initiating capacity and chemoresistance. The extracellular matrix , a major structural component of the tumor microenvironment, is a highly dynamic structure and increasing evidence suggests that ECM proteins establish a physical and biochemical niche for CSCs. In cancer, abnormal ECM dynamics occur due to disrupted balance between ECM synthesis and secretion and altered expression of matrix-remodeling enzymes. Tumor-derived ECM is biochemically distinct in its composition and is stiffer compared to normal ECM. In this review, we will provide a brief overview of how different components of the ECM modulate CSC properties then discuss how physical, mechanical, and biochemical cues from the ECM drive cancer stemness. Given the fact that current CSC targeting therapies face many challenges, a better understanding of CSC-ECM interactions will be crucial to identify more effective therapeutic strategies to eliminate CSCs.
Permeability Of The Extracellular Matrix
The extracellular matrix is not solid and amorphous but contains numerous aqueous pores, and like other polysaccharide networks , it acts as a sieve through which water and polar molecules can easily diffuse when the extracellular matrix is hydrated . The extracellular matrix becomes limiting to water movement when it is dehydrated, and evaporation is the driving force for water movement . The average sizes of pores in semidehydrated extracellular matrices were originally estimated to be approximately 35 nm in diameter , whereas those in hydrated extracellular matrices were estimated to be approximately twice as large . Therefore, when the extracellular matrix is hydrated, its permeability is typically greater than that of the plasma membrane and can be ignored as a first approximation when measuring the permeation of solutes into or out of the cell .
The original experiments to determine pore size of the extracellular matrix were done in 100300 mol/m3 solutions that contained solutes that were either small enough to pass through the extracellular matrix and plasmolyze the cell or were too large to pass through the extracellular matrix and caused it to crinkle . Either way, the 0.1- to 0.3-M solutions dehydrated the extracellular matrix during the experiment and thus provided a minimum estimate of the average pore size .
K.M. Reiser, in, 2007
You May Like: How To Avoid Parallax Error In Physics
Ecm And Gene Expression
Image: gene expression
The extracellular matrix has a role in gene expression. Genes are activated by the ECM and other cells, as well as the extracellular proteins.
There are a few different Extracellular matrix types: collagen, elastin, and proteoglycans.
Proteoglycans are proteins that have sugar chains attached to them. They help cells bind together, and they also regulate cell growth, differentiation, and movement.
Collagens are found all over the body, from internal support structures to skin tissue. They provide strength and structure in tissues.
Protein elastin is found in places that need to stretch, such as skin and lungs. They help the cells maintain their elasticity so that they can return to their original shape.
Gene expression is the process of producing RNA from DNA. It is a complicated process that involves multiple steps.
The ECM triggers gene expression and provides signals to the cells to control the process.
Building An Ecm Atlas
The interrogation of MatrisomeDB by selecting an entire matrisome category, for example the proteoglycans, results in the generation of a hierarchically-clustered tissue distribution heatmap of all 30 proteoglycans detected and thus can constitute the Proteoglycan atlas . Similarly, the ECM atlas of any of the tissues or any of the organisms included in this database can be generated.
Also Check: How To Study For Biology Keystone Exam
What Makes The Extracellular Matrix
The extracellular matrix is a collection of macromolecules and polysaccharides secreted outside the cell membrane.
The components of the ECM are arranged in an organized fashion to offer cell protection, signalling, and structural support.
The ECM consists mostly of collagen fibres made from amino acid chains. These chains branch out into other structures like proteoglycans, glycoproteins, and other ECM proteins.
Ecm Main Structural And Functional Components At A Glance
Extracellular matrices are multiplicate well-organized 3-dimensional architectural networks with critical structural and functional roles in tissue organization and remodeling and in the regulation of cellular processes ]. The building blocks of these ultrastructures are the collagens, proteoglycans and glycosaminoglycans , elastin and elastic fibers, laminins, fibronectin, and other proteins/glycoproteins such as matricellular proteins ]. ECMs operate as communication liaisons between the cells in organs and tissues by coordinating multiple signaling inside-out or outside-in commands ]. As a consequence, ECMs guide tissue morphogenesis, development and homeostasis, through the regulation of cellular physiology, growth, survival, differentiation, and adhesion. ECMs undergo extensive remodeling during pathological conditions acting as key players driving disease progression ]. Specific ECM phenotypes configure the different tissues to meet the requirements for optimal tissue functions ]. Nevertheless, the formulation of ECMs can constantly be adapted according to biochemical or mechanical signals, resulting in a fine-tuned vivid ECM remodeling procedure ]. Schematic representations of the main ECM networks and their composition in the various tissues are given in Fig..
Fig. 1
A short introduction to each one of the main macromolecular components that construct the core of the ECM networks is given below.
Read Also: What Are The 4 Major Goals Of Psychology
The Animal Extracellular Matrix
Image: Animal extracellular matrix
The animal extracellular matrix is made up of structural and signalling molecules and proteoglycans that tell cells to turn off immune responses.
Inside the ECM, some proteins bind to specific receptors on cells and initiate signalling cascades to promote homeostasis.
The ECM also drapes around epithelial cells and creates a physical barrier to prevent infection by viruses or bacteria. The animal ECM consists of the basement membrane and the interstitial matrix.
Stressed by a biology assignment? Our ivy league writers are ready to take the stress from you at a friendly fee. Follow the steps below:
- Describe your assignment and fill the details
- Confirm payment
The Structure And Function Of Extracellular Matrix
An essential part of the holding capacity of tissues is the extracellular area. The extracellular region is primarily occupied by a complicated network of macromolecules constituent called as extracellular matrix . The composition of ECM is varied, depends on the species and also developing or ground molecules . Commonly, the ECM is composed of three major classes of biomolecules there are glycosaminoglycans , linked to a protein known as the proteoglycans, and also fibrous proteins, including collagen, elastin, fibronectin, vitronectin, and laminin.
Figure 1.
The structure of the extracellular matrix. The ECM mainly contained collagen fibers. There are also some glycoproteins as an adhesion molecule, such as integrin family fibronectin and laminin, which conduct cell attachments to the ECM by binding to collagen in the ECM and integrin. The intracellular part of integrin highly associated with the cytoskeleton thus may promote to anchoring the cell. In the end, there are various proteoglycans in the ECM that act as primary proteins and are profoundly modified by the addition of sugars.
Figure 2.
The extracellular matrix of hyaline cartilage found in abundant collagen fibril and proteoglycan aggregates. The chemical analysis of the ground substance reveals that it contains a few glycoproteins and a high concentration of three types of glycosaminoglycans: hyaluronic acid, chondroitin sulfate, and keratan sulfate. Adapted from Crammer and Bakkum .
Figure 3.
Recommended Reading: How Much Do Physics Professors Make
Bits And Pieces Molecular Composition Of The Ecm
The ECM is composed of two main classes of macromolecules: proteoglycans and fibrous proteins . The main fibrous ECM proteins are collagens, elastins, fibronectins and laminins . PGs fill the majority of the extracellular interstitial space within the tissue in the form of a hydrated gel . PGs have a wide variety of functions that reflect their unique buffering, hydration, binding and force-resistance properties. For example, in the kidney glomerular BM, perlecan has a role in glomerular filtration . By constrast, in ductal epithelial tissues, decorin, biglycan and lumican associate with collagen fibers to generate a molecular structure within the ECM that is essential for mechanical buffering and hydration and that, by binding GFs, provides an easy, enzymatically accessible repository for these factors .
Box 1. Structure and function of proteoglycans
Box 2. Collagen and fibronectin synthesis
FN is secreted as a dimer joined by two C-terminal disulfide bonds and has several binding sites to other FN dimers, to collagen, to heparin and also to cell-surface integrin receptors . Cell-surface binding of the soluble FN dimer is essential for its assembly into longer fibrils. Moreover, cell contraction through the actomyosin cytoskeleton and the resulting integrin clustering promotes FNâfibril assembly by exposing cryptic binding sites, thus allowing them to bind one another .
Extracellular Matrix In Plants
Plant cells are tesselated to form tissues. The cell wall is the relatively rigid structure surrounding the plant cell. The cell wall provides lateral strength to resist osmotic turgor pressure, but is flexible enough to allow cell growth when needed it also serves as a medium for intercellular communication. The cell wall comprises multiple laminate layers of cellulose microfibrils embedded in a matrix of glycoproteins such as hemicellulose, pectin, and extensin. The components of the glycoprotein matrix help cell walls of adjacent plant cells to bind to each other. The selective permeability of the cell wall is chiefly governed by pectins in the glycoprotein matrix. Plasmodesmata are pores that traverse the cell walls of adjacent plant cells. These channels are tightly regulated and selectively allow molecules of specific sizes to pass between cells.
Read Also: Geometry Dash The Secret Box
Extracellular Matrix Proteins And Cellular And Molecular Response In The Peripheral Nervous System
In addition to the role of neurotropic factors and calcium modulation in promoting regeneration after axotomy, the matrix proteins of the basal lamina and endoneurium also aid in this process. Specifically, the basal lamina contains laminin B, laminin 2, and laminin 10 that have neurite-promoting properties in the distal nerve stump. Once the process of axonal regeneration is initiated, glial growth factor mediates the maturation of immature peripheral nerve cells.
Iwen Wu, Jennifer Elisseeff, in, 2014
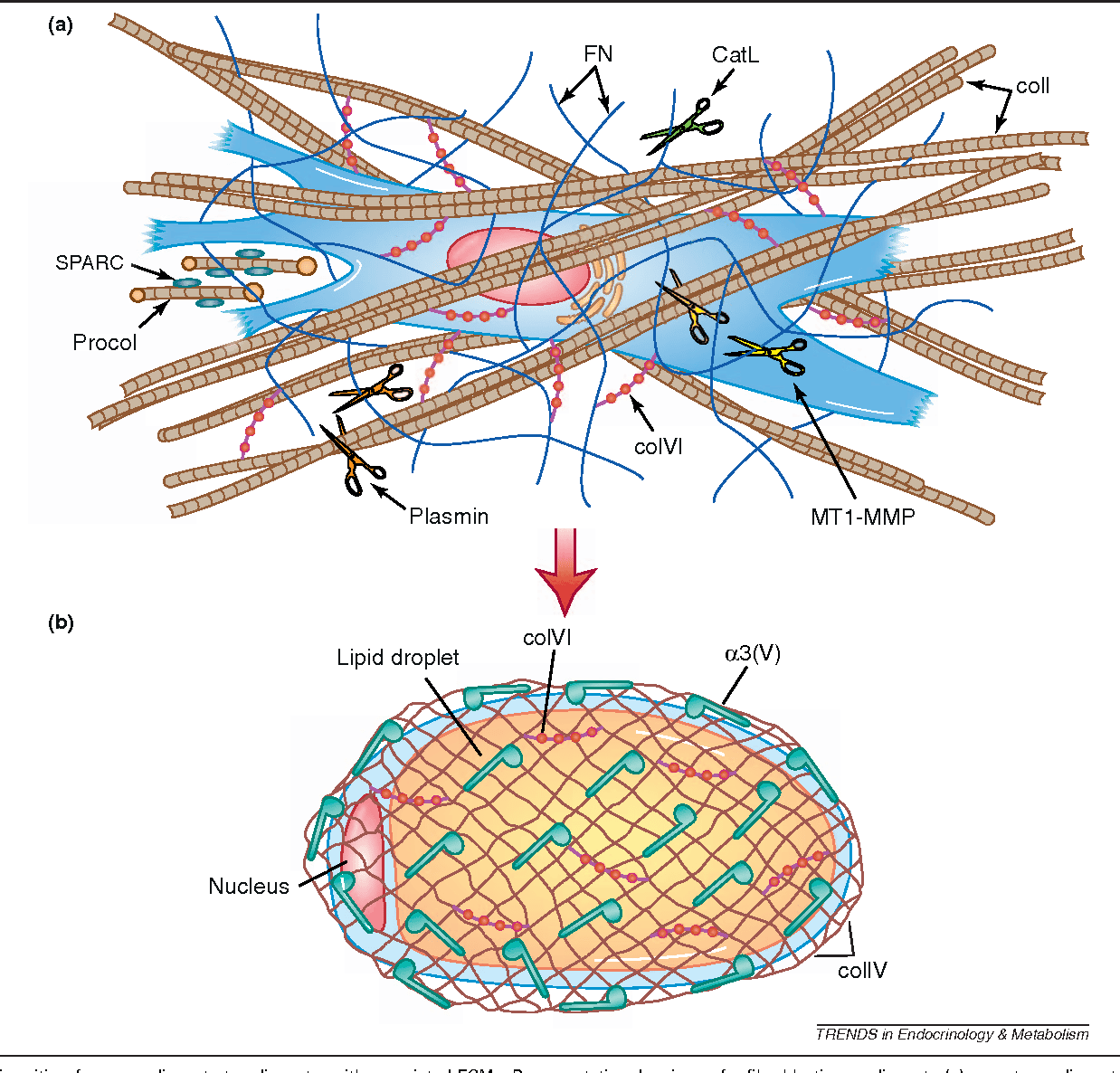
Collagen Forming Beaded Filaments
Collagen VI forms 100-nm periodic end-to-end beaded filaments ] and contains vWF A and Kunitz family of serine protease inhibitors domains. It plays a key role in skeletal muscle and mutations in the COL6A1, COL6A2, and COL6A3 genes encoding collagen VI chains lead to congenital muscular dystrophies ]. Collagen VI is required for the structural and functional integrity of the neuromuscular junction ] and is a component of the peripheral and CNS ].
Like collagen VI, collagen XXVIII contains vWF A and Kunitz family of serine protease inhibitors domains ]. It is located in the BMs around Schwann cells and is associated with nonmyelinated regions of the peripheral nervous system such as the nodes of Ranvier ] but it has not been reported to form specific supramolecular assemblies so far. The C-terminal Kunitz domain is collagen XXVIII is often proteolytically processed in zebrafish ]. The lack of collagen XXVIII in mice causes age-related insufficiency in retinal pigment epithelium proteostasis in mice ]. COL28A1 gene is one of the prognostic feature genes identified in glioblastoma multiforme, the most common type of brain cancer ].
Read Also: Pearson Texas Algebra 1 Form K
Structural Organization And Functions Of Basement Membrane And Connective Tissue
Underlying the BM is the so-called interstitial connective tissue that encloses, separates, and supports tissues. Its ECM composition can be variable to create specific environments for distinct tissue function and loose, dense, and specialized connective tissues can be distinguished. Collagens, mainly type I, but also type III resist to tensional loads and elastin may further support additional tissue stretching. On the contrary PGs, also abundant in these ECMs, opposite compression forces. Moreover, specialized matrix components such as PGs and proteins boost the tissue mechanochemical properties ]. Bone and cartilage consist of specialized ECM. Cartilage ECM is composed mainly of a viscous gel enriched by mostly avascular PG aggregates, while bones consist of rigid mineralized and vascularized ECM.
ECM composition in cartilage
ECM in bone
ECM organization in cornea
How Does The Process Of Tissue Regeneration Happen
The process of tissue regeneration can happen in a few different ways, such as by scar formation or through the addition of new cells. Scar formation is the process of forming a fibrous network over the site where tissue is lost.
The new cells are either added through cell migration and proliferation or by differentiation of cells that already exist in the area. Scar tissue is not as strong or resilient as native tissue, but it can still be functional.
There are two types of scars: hypertrophic and atrophic. Hypertrophic scars are raised, red, thickened areas that can lead to keloids. Atrophic scars are depressions in the skin and can be caused by burns, acne, or surgery.
Fibroblasts determine how a wound heals. They produce collagen that links to the wound and then fills in the gap where the tissue was lost. They can also be used in tissue engineering.
Tissue engineering is a process of rebuilding damaged tissue. It is done using synthetic biological and natural materials that closely resemble the original cells or tissues being repaired.
Different cells have different roles in wound healing:
Tired of reading? We get it. Let our ivy league writers take the stress of that assignment from you at an affordable fee. Additionally, you get the following premium features for free!
Recommended Reading: How To Calculate Half Life Chemistry
Ecm Modulates Immune Surveillance In Csc Microenvironment
Extracellular matrix can profoundly influence recruitment of immune cells into the tumor microenvironment. CSCs can evade immune surveillance by altering this microenvironment to favor their survival. For example, ECM drives the activation of pro-survival pathways such as PI3K/AKT, which has been shown to facilitate immune evasion in CSCs . ECM proteins can recruit immunosuppressive cells such as tumor-associated macrophages and regulatory T cells that have been known to promote CSC survival, while simultaneously blocking the recruitment of antitumorigenic immune cells such as cytotoxic T cells . In addition, the ECM composition can dramatically modulate the activation state of the tumor infiltrating immune cells. For instance, a stiff collagen-rich or POSTN-rich ECM allows macrophage polarization to a pro-tumorigenic M2 phenotype . Following recruitment, the M2 macrophages activate several CSC survival signaling pathways including Src, NF-B , STAT3/SOX2 and Hedgehog . ECM can also impair proliferation and activation of T cells, that are required for capturing and killing CSCs . A collagen-rich ECM can inhibit T-cell proliferation and activation through type I collagen-dependent fusion of LAIR receptors in addition to sequestering growth factors required for T cell proliferation . Furthermore, TAMs and neutrophils that can selectively reorganize the ECM to promote malignant growth of cancers are preferentially recruited to the microenvironment.