Placing Electrons Appropriately Between Atoms
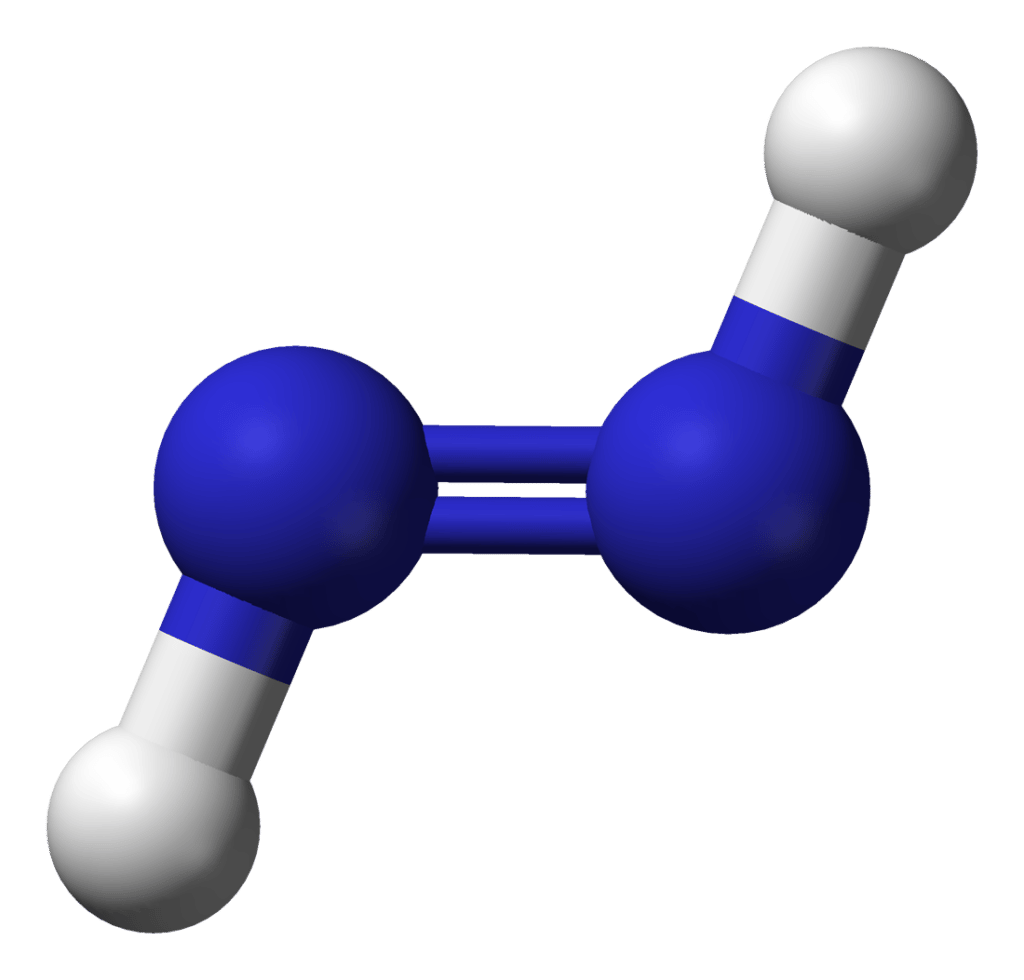
In this structure two types of bonds are observed, one is H-N formed by sharing of one pair of electrons between hydrogen and Nitrogen. Resulting is a single bond. The other bond is N-N bond where two electron pairs are shared between the two nitrogen. Resulting bond is a double bond.
N2H2 lewis structure
Is N2h2 Ionic Or Covalent
The bonding in covalent compounds is due to sharing of electrons and in ionic it is due to charges formed between metal and a non-metal. Let us check for N2H2 .
N2H2 is covalent. The bond formed in this molecule is as a result of sharing electron pairs between the atoms. Also the bond formation occurs between two non metals, confirming that the bond is covalent type.
N2H2 is one of the hydride of nitrogen which exists in gas form. The type of bonding observed in this molecule is covalent and is acidic in nature.
Check Whether The Molecule Is Symmetric Or Not
Have a look at this 3D structure of N2H2. The two Nitrogen atoms are at the center and it is surrounded by Hydrogen atoms .
It also has one lone pair on each Nitrogen atom .
Due to the lone pair on the nitrogen atoms , its molecular geometry becomes asymmetric.
Because of this, there are positive and negative poles of charges on the overall molecule of N2H2.
Hence, the N2H2 molecule is a polar molecule.
I hope you have understood the reason behind the polar nature of N2H2 molecule.
See the polarity of other molecules to make your concepts clear:
Also Check: What Are Halogens In Chemistry
Check Whether The Central Atom Has Octet Or Not If It Does Not Have An Octet Then Convert The Lone Pair Into Double Bond Or Triple Bond
In this step, we have to check whether the central atoms have an octet or not.
In simple words, we have to check whether the central Nitrogen atoms has 8 electrons or not.
As you can see from the above image, one of the nitrogen atoms has only 6 electrons. So it does not fulfill the octet rule.
Now, in order to fulfill the octet of this nitrogen atom, we have to convert the lone pair into a double bond.
Now you can see from the above image that both the central nitrogen atoms are having 8 electrons. So they fulfill the octet rule and both the nitrogen atoms are stable.
Why Is There No Double Bond In The N2h4 Lewis Dot Structure
As you closely see the N2H4 lewis structure, hydrogen can occupy only two electrons in its outer shell, which means hydrogen can share only two electrons. So, there is no point we can use a double bond with hydrogen since a double bond contains a total of 4 electrons.
Now lets talk about the N-N bond, each nitrogen has three single bonds and one lone pair. If we convert the lone pair into a covalent bond then nitrogen shares four bonds.
But the problem is if a double bond is present in the N2H4 dot structure, then it becomes unstable. As nitrogen atoms will get some formal charge.
Lewis structure is most stable when the formal charge is close to zero.
Thats why there is no need to make any double or triple bond as we already got our best and most stable N2H4 lewis structure with zero formal charges.
Also Read:
Read Also: What Is Elliptical Orbit In Geography
Lewis Structure Of N2h4
Lewis dot diagram or electron dot structure is the pictorial representation of the molecular formula of a compound along with its electrons that are represented as dots.
These structures are named after American chemist Gilbert Newton Lewis who introduced them in 1916.
Lewis structures illustrate the chemical bonding between different atoms of a molecule and also the number of lone pairs of electrons present in that molecule.
These are the representation of the electronic structure of the molecule and its atomic bonding where each dot depicts an electron and two dots between the atoms symbolize a bond.
The Lewis structure of N2H4 is given below.
We already know that only the valence electrons of an atom participate in chemical bonding to satisfy the octet for that atom. In the case of N2H4 nitrogen has five electrons while hydrogen has only one valence electron.
Here, this must be noted that the octet rule does not apply to hydrogen which becomes stable with two electrons.
Therefore, each nitrogen atom forms a single bond with two hydrogen atoms and the other nitrogen atom, thus, satisfying the octet rule for all the participating atoms.
N2h4 Lewis Dot Structure
Yes, we completed the octet of both atoms and also used all available valence electrons.
I think we completed the lewis dot structure of N2H4? No, we need one more step to verify the stability of the above structure with the help of the formal charge concept.
6. Check the stability with the help of a formal charge concept
A formal charge is the charge assigned to an atom in a molecule, assuming that electrons in all chemical bonds are shared equally between atoms.
The structure with the formal charge close to zero or zero is the best and most stable lewis structure.
To calculate the formal charge on an atom. Use the formula given below-
Formal charge =
We will calculate the formal charge on the individual atoms of the N2H4 lewis structure.
For nitrogen atom:
Valence electrons of nitrogen = 5
Lone pair electrons on nitrogen = 2
Shared pair electrons = 6
= 0 formal charge on the nitrogen atom
For hydrogen atom
Valence electrons of hydrogen = 1
Lone pair on hydrogen = 0
Shared pair electrons = 2
= o formal charge on the hydrogen atom
Hence, the overall formal charge in the N2H4 lewis structure is zero.
Therefore, we got our best lewis diagram.
Also check
Read Also: College Algebra With Intermediate Algebra
N2h2 Lewis Structure Polar Or Nonpolar
N2H2 is a Lewis acid. It has two hydrogen atoms, one on each nitrogen atom. One of the hydrogens can be either polar or nonpolar . When it is polar, it will have an uneven distribution of positive and negative charges that attract electrons in order to create stability. This type of molecule is called a dipole. A non-polar hydrogen atom does not have any charge at all and will only attract other neutral molecules. N2H2s polarity determines whether it is considered to be acidic or basic in nature because these properties are based on their ability to donate protons in chemical reactions with water molecules in order to form H3O+.
What Is Vsepr Theory
We are all aware of the fact that electrons are negatively charged particles that are present on the shells or orbitals surrounding atomic nuclei. Now, having negative charges make them repulsive since they are like-charged particles.
So, according to VSEPR theory, the repulsion that is created by the electron cloud surrounding the atomic nuclei needs to be minimized for maintaining stability and balance inside a molecule.
VSEPR model predicts the 3D molecular geometry or shape of a chemical compound.
So, how does an N2H2 molecule appear in 3D?
You May Like: What Is Genetics In Biology
How Are The Shapes And Geometry Of N2h2 And N2h4 Different
In N2H2, 2 hydrogen atoms are bonded to the central N atoms, one on each side while in N2H4, a total of 4 hydrogen atoms are bonded to the central N atoms, two on each side.
The central N-atoms are bonded to each other via a double covalent bond in N2H2 in contrast to a single bond present between the central N-atoms in N2H4.
However, there is one lone pair on each N atom at the center in both N2H2 and N2H4
Considering these differences, the AXN notation for the N2H2 molecule is AX2N1 thus according to the VSEPR concept, it has a bent shape and molecular geometry while its ideal electron geometry is trigonal planar.
The AXN notation for the N2H4 molecule is AX3N1 consequently it has a trigonal pyramidal shape and molecular geometry while its electron geometry is tetrahedral.
Also Read:-
N2h2 Lewis Structure Molecular Geometry Hybridization And Mo Diagram
Dinitrogen dihydride has the chemical formula of N2H2. This compound is most commonly known as diazene or diimide. It is a yellowish-colored gas having both cis and trans isomers. It can be prepared from the decarboxylation of azodicarboxylic acid 2 ).
Also, the application of air or H2O2 leads to oxidation of hydrazine which results in the formation of N2H2.
Below is the chemical equation of preparation of Dinitrogen Dihydride/Diazene/Diimide
2 > 2 + 2 CO2
It melts at 193K and has a molar mass of 30.03 g/mol. It has a density of around 1.2 g/cc. Azo and diazo groups are substituents from diazene.
N2H2 has a lot of uses in organic synthesis. For example, it helps in the hydrogenation of alkenes and alkynes. Other than this, it also aids in the selective reduction of branched acyclic hydrocarbons.
Read Also: What Is Neuroticism In Psychology
N2h4 Lewis Structure Molecular Structure Hybridization Bond Angle And Shape
Hydrazine is an inorganic pnictogen with the chemical formula N2H4. It is a colorless liquid with an Ammonia-like odor.
Hydrazine is highly flammable and toxic to human beings, producing seizure-like symptoms. It is primarily used as a foaming agent but also finds application in pesticides, airbags, pharmaceuticals, and rocket propulsion. It is also a potent reducing agent that undergoes explosive hypergolic reactions to power rockets. This inherent property also dictates its behavior as an oxygen scavenger, as it reacts with metal oxides to significantly reverse corrosion effects.
The creation of the single-bonded Nitrogen molecule is a critical step in producing Hydrazine. The Raschig process is most commonly employed to manufacture Hydrazine on a large scale. Ammonia is oxidized in the presence of Sodium Hypochlorite to form Hydrogen Chloride and Hydrazine.
Nitrogen and Oxygen are released when Hydrazine undergoes Oxygen-induced combustion. As a potent reducing agent, it reacts with metal salts and oxides to reverse corrosion effects. Hydrazine forms salts when treated with mineral acids. Hydrazine sulfate use is extensive in the pharmaceutical industry.
Lets understand Hydrazine better. Some of its properties are given in the table below:
| Name of the molecule | |
| + = 14 valence electrons | |
| Hybridization of the central atom | sp3 |
What Is The Molecular Geometry Of N2h2 Lewis Structure
The Lewis structure of N2h2 is an example of a molecule that can be described by resonance. This means that the nitrogen atom has 6 valence electrons, but it only shares 4 electron pairs with hydrogen atoms. The remaining two are shared between the two nitrogen atoms in order to give each atom 8 valence electrons. Atoms will try to share their electron pairs equally so this is considered stable.
The Lewis structure is a graphical representation of the electron configuration for atoms. It represents the valence shell electrons around an atom, which are shared with other atoms to form chemical bonds. The Lewis model can be used to predict molecular geometry based on bond angles and number of non-bonding pairs. The molecular geometry of N2h2 lewis structure is tetrahedral due to its four nitrogen-hydrogen single bonds arranged in a tetrahedral shape, with each hydrogen attached by covalent bonding to one nitrogen atom at 109° angles from each other.
Read Also: What Does Sustainable Mean In Geography
Final Step Check The Stability Of Lewis Structure By Calculating The Formal Charge On Each Atom
Now, you have come to the final step and here you have to check the formal charge on nitrogen atoms as well as hydrogen atoms .
For that, you need to remember the formula of formal charge
Formal charge = Valence electrons Nonbonding electrons /2
- For Nitrogen:Valence electrons = 5 Nonbonding electrons = 2
- Valence electron = 1 Nonbonding electrons = 0
Follow Some Steps For Drawing The Lewis Dot Structure Of N2h4
1. Count total valence electron in N2H4
As we know, lewiss structure is a representation of the valence electron in a molecule. So, in the first step, we have to count how many valence electrons are available for N2H4.
I assume that you definitely know how to find the valence electron of an atom. The valence electron of an atom is equal to the periodic group number of that atom.
So, nitrogen belongs to the 15th periodic group, and hydrogen to the 1st group. Therefore, the valence electron for nitrogen is 5 and for hydrogen, it is 1.
Total number of the valence electron in Nitrogen = 5
Total number of the valence electrons in hydrogen = 1
Total number of valence electron available for the N2H4 lewis structure = 5 + 1 = 14 valence electrons
2. Find the least electronegative atom and placed it at center
Now its time to find the central atom of the N2H4 molecule. It doesnt matter which atom is more or less electronegative, if hydrogen atoms are there in a molecule then it always goes outside in the lewis diagram.
Because hydrogen only needs two-electron or one single bond to complete the outer shell. So, for N2H4, put away hydrogen outside and nitrogen as a central atom in the lewis diagram.
3. Connect outer atoms to central atom with a single bond
In this step, we need to connect every outer atom to the central atom with the help of a single bond. And make sure you must connect both nitrogens with a single bond also.
= 4 valence electrons
Recommended Reading: Application Of Linear Algebra In Daily Life
Complete The Octet On Outside Atoms If The Valence Electrons Are Left Then Put The Valence Electrons Pair On The Central Atom
Dont worry, Ill explain!
In the Lewis structure of N2H2, the outer atoms are hydrogen atoms as well as a nitrogen atom.
So now, you have to check whether these hydrogen atoms are forming a duplet or not! .
Also, the outer nitrogen atom forms an octet.
You can see in the above image that both the hydrogen atoms form a duplet and a nitrogen atom also forms an octet.
Also, only 10 valence electrons of N2H2 molecule are used in the above structure.
But there are total 12 valence electrons in N2H2 molecule .
So the number of electrons left to be kept on the central nitrogen atoms = 12 10 = 2.
So lets keep these two electrons on the central nitrogen atom.
Now, lets move to the next step.
Deciding The Least Electronegative Atom
An important step where the atom with least electronegativity is found out. So the atom having least electronegativity will be placed at the centre while drawing the structure. And other atoms will bond with it by sharing pairs of electrons. The atom here that has to be placed in the centre is Nitrogen.
Also Check: What Is Pollen Grain In Biology
N2h2 Lewis Structure Molecular Geometry Hybridization Bond Angle And Shape
N2H2 is a chemical formula for a Diazene molecule which is also known as Nitrogen Hydride. It is the conjugate acid of a diazenide. The molecule is made up of two hydrogen atoms and two nitrogen atoms. We will first learn the Lewis structure of this molecule to understand its physical and chemical properties better. The structure will help know the arrangement of atoms, bond formation, and shape of the molecule.
| Name of molecule | |
| No of Valence Electrons in the molecule | 12 |
| Molecular Geometry of N2H2 | Trigonal Pyramidal |
We will now look at the N2H2 Lewis structure. But for knowing the Lewis structure of any molecule, we first need to know the total number of valence electrons.
Contents
Lewis Structure Of N2h2
The first step towards sketching the Lewis Structure of any molecule is to understand the concept of valency and calculation of valence electrons.
Valency is defined as the combining capacity of an atom for bond formation. This is indicated usually by the number of electrons in the outermost shell, also known as the valence shell.
Elements belonging to the same periodic table group will have the same number of valence electrons.
In the case of N2H2, a single molecule has two atoms of nitrogen and two atoms of hydrogen.
Let us look at the periodic table.
Nitrogen belongs to group 15 and has 5 valence electrons.
Hydrogen belongs to group 1 and has 1 valence electron.
Total number of valence electrons in N2H2 = 5*2 + 1*2 = 12.
Now, we have to identify the central atom in the molecule.
Here, we can see the Pauling Electronegativity chart. The general rule is to consider the element having the least electronegativity value as the central atom.
But an exception to this rule is the hydrogen atom which prefers to sit outside.
So, our rough sketch looks like this:
In Lewis Structure, we use the electron dot notations, i.e., we denote the valence shell electrons of the constituent atoms via dot symbols.
Now, here comes the octet fulfillment or the octet rule.
Exception: Hydrogen tends to fulfill 2 electrons in its outer shell since it follows a Helium configuration.
To fulfill the octet of the other N atom, we will have the Lewis Structure modified as:
Also Check: What Are All The Laws Of Physics
N2h2 Lewis Structure Angle
The shape of the structure helps in predicting the bond angle. Let us find out the bond angle in diimide.
The N2H2 lewis structure angle is 120°. The reason for this is the geometry is of the type AX2E having 1 lone pair of electrons, so there will be repulsion. This makes the structure into a bent form where the angle of bonds comes down to 120°.
Hybridization Of N2h2 Molecule

Hybridization is the concept of mixing atomic orbitals and forming new hybrid orbitals, which in turn influences molecular geometry and bonding properties. It is an integral part of the valence bond theory.
N2H2 has an sp2 hybridization as it has three sigma bonds and one lone pair of electrons on central two nitrogen atoms. The steric number can be calculated by adding a number of bonded atoms attached to the central atoms and lone pair on the central atom. Hence, the steric number comes out three which indicates sp2 hybridization.
Noteonly sigma bond takes part in hybridization but pi bond doesnt take part in hybridization.
Now steric number in N2H2 molecule = Central one nitrogen is attached to one hydrogen and one N-H + central one nitrogen atom has one lone pair.
Here, steric number of center carbon atom = 3+0
Hence, the steric number of central carbon atoms in N2H2 molecule is three which shows sp2 hybridization.
We can also determine hybridization of molecules first of all to know the electronic configuration of atoms present in molecule.
Electronic configuration of carbon, N: 1s2 2s2 2p3
Electronic configuration of hydrogen, H: 1s2
Sigma-bond: single bond formed by sigma bond
Pi-bond: after the sigma bond another addition of multiple bonds is formed by pi bond.
Read Also: How Do Crumple Zones Work Physics