Lipid Composition Of The Plasma Membrane
The most direct evidence that the fraction released can be altered was presented by both the Amatore and Ewing groups who showed that the amount of neurotransmitter released per event can be quickly altered by influencing the lipid composition of the plasma membrane . In one case, they found that a short-term incubation of PC12 cells with the inverted cone shaped lipid lysophosphatidylcholine increased the average amount of dopamine released per exocytosis event while incubation with the cone shaped phosphatidylethanolamine decreased it. In the other case, a rapid alteration of amount released per event was demonstrated by Amatore and coworkers who used LPC and arachidonic acid , which displays a cone shape similar to that of PE, to study the effect of membrane lipid composition on the dynamics of exocytosis from bovine chromaffin cells. They found that LPC facilitates catecholamine release while AA inhibits the exocytotic processes. They also observed that the detected amount of neurotransmitters in the presence of LPC was larger than under control conditions, while the opposite trend is observed with AA .
Basic Mechanism Of Exocytosis
- The first step of Exocytosis involves the transportation of vesicles containing molecules from the inside of the cell to the cell membrane.
- After that, the vesicle gets attached to the cell membrane.
- During the fusion of the vesicle membrane with the cell membrane, the vesicle contents are released outside the cell.
Transient But Not Prolonged Calcium Increase Triggers Endocytosis
Increasing the duration of APlike trains or depolarization trains prolongs endocytosis at goldfish retinal bipolar nerve terminals , frog neuromuscular junctions , hippocampal synapses , calyces , and chromaffin cells . For three reasons, these observations are not in conflict with the finding that calcium influx at the micro/nanodomain triggers endocytosis. First, calcium from the micro/nanodomain dissipates within milliseconds after action potentials, whereas action potentials are often repeated at an interval of ~50 ms or longer. Thus, repetitive action potentials at low to intermediate frequencies may not substantially increase the transient calcium concentration at the micro/nanodomain . Second, repeated stimulation causes more exocytosis, which may saturate the endocytic capacity, resulting in apparently slower endocytosis . Third, repeated stimulation increases global calcium concentration to the submicromolar range for a prolonged period of time . Prolonged dialysis of calcium at 1 M or more in goldfish retinal nerve terminals and chromaffin cells inhibits endocytosis . Thus, transient, but not prolonged, calcium increase triggers endocytosis.
Read Also: Cryptic Quiz Page 148 Answers
What Is Exocytosis With An Example
Exocytosis is the process by which cells eliminate large substances such as hormones, digestive enzymes, and undigested waste products from within the cell to its exterior through the plasma membrane. Examples of exocytosis include: Secretion of antibodies, enzymes and peptide hormones from different cells
Vesicle Function In Endocytosis And Exocytosis
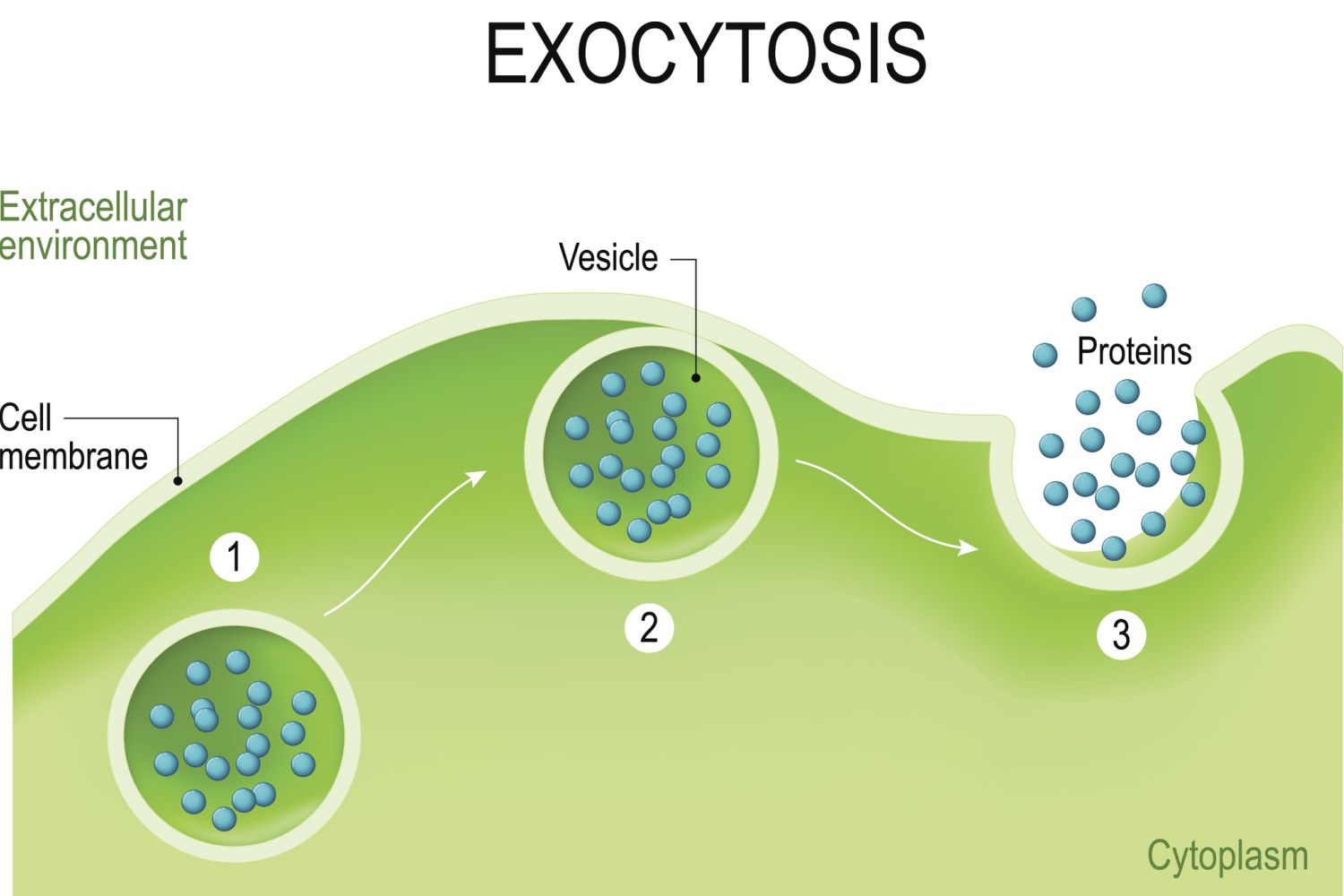
During bulk transport, larger substances or large packages of small molecules are transported through the cell membrane, also known as the plasma membrane, by way of vesicles think of vesicles as little membrane sacs that can fuse with the cell membrane.Cell membranes are comprised of a lipid bilayer. The walls of vesicles are also made up of a lipid bilayer, which is why they are capable of fusing with the cell membrane. This fusion between vesicles and the plasma membrane facilitates bulk transport both into and out of the cell.
You May Like: Geometry Of Ccl4
The Total Content Of A Secretory Vesicle Is Greater Than The Amount Released
Fig. 5.Electrochemical cytometry with a three layer PDMS device for the end-column lysis and electrochemical detection of vesicles separated by capillary electrophoresis . Schematic of device . Bright field and confocal fluorescence images of the device.
Fig. 6.Amperometric quantification of catecholamine amounts released from during exocytosis in PC12 cells with amperometry and in vesicles with electrochemical cytometry . Representative amperometric trace resulting from exocytotic release at intact cells. Red arrow indicates where an elevated K+ application was used to induce exocytosis. Electrochemical cytometry of individual vesicles. Normalized frequency histogram of the vesicular catecholamine amounts quantified from intact cells that underwent stimulated exocytosis versus isolated vesicles investigated with electrochemical cytometry.
Fig. 7.The electrochemical response to single adrenal chromaffin vesicles filled with catecholamine hormones as they are adsorbed and rupture on a 33 µm diameter disk-shaped carbon electrode. Representative trace from a suspension of chromaffin cell vesicles. A 5-s baseline at 0 mV versus Ag|AgCl in the presence of vesicles. Expanded view of current transients. The pink squares represent the Imax of all peak candidate peaks submitted for further analysis. The green line represents the root-mean-square and the red line five times the RMS of the baseline noise .
What Is Exocytosis And Endocytosis In Biology
-
In this fluid what is exocytosis and endocytosis in biology post we have briefly explained about exocytosis and endocytosis in biological cell membrane.
-
Certain particles or molecules are too big to traverse the plasma membrane, or pass through a transporter. Cells use two different active transport mechanisms to move those macromolecules to and out of cells.
-
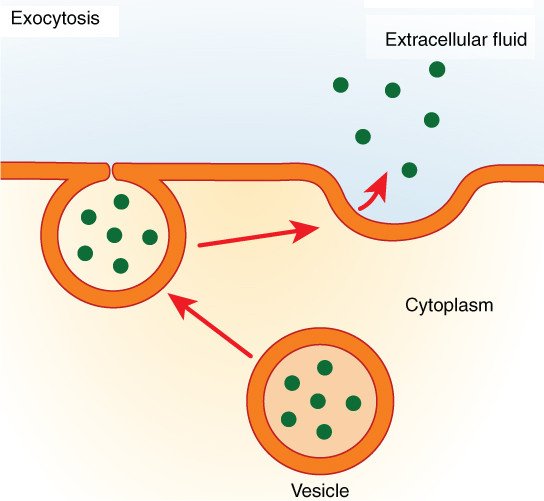
Vesicles or other structures within the cytoplasm transport macromolecules or other large particles over the membrane. There are two kinds of vesicle transport: exocytosis as well as endocytosis . Both of these are transport-related processes that are active, which require energy.
Exocytosis and Endocytosis Process: Illustration of the two types of vesicle transport, exocytosis and endocytosis. Image Source:
Read Also: Who Are Paris Jackson’s Biological Parents
Exocytosis Definition Types Steps Examples
Exocytosis is a type of process, in which involves the movement of materials from the inside of a cell to the exterior…
MN EditorsThis article writter by MN Editors on October 18, 2021
Microbiology Notes is an educational niche blog related to microbiology and different branches of biology.
THIS BLOG INCLUDES:
A Timeline For Exocytosis
Ben Short A timeline for exocytosis. J Cell Biol 20 July 2015 210 : 173. doi:
Exocytosis is essential not only for secreting proteins outside of the cell, but also for supporting cell growth . Secretory vesicles are delivered to the plasma membrane and tethered there for short periods before undergoing membrane fusion to release their contents. Researchers have identified and characterized many of the proteins involved in these processes, but precisely when, and in which order, they all act remains unclear. By tracking the tethering and fusion of individual secretory vesicles, Donovan and Bretscher sketch out a timeline for exocytosis in budding yeast .
Yeast secretory vesicles carry the Rab GTPase Sec4p, which, once activated by its guanine nucleotide exchange factor Sec2p, recruits a protein complex called the exocyst and binds and activates the myosin motor Myo2p, which transports the vesicle along actin cables into the bud. The exocyst then tethers the vesicle to the plasma membrane before Sec4p is inactivated and SNARE proteins and regulatory factors such as Sec1p stimulate membrane fusion.
The vesicles remained tethered for a well-defined time of 18 seconds.
Read Also: 6 Major Branches Of Chemistry
The Total Content Of A Secretory Vesicle Is Greater Than The Amount Released In Pc12 Cells
The amount released from individual vesicles containing electroactive neurotransmitters such as dopamine can be measured through electrochemical methods. Work in the Ewing laboratory compared conventional amerometric measurements of exocytosis with electrochemical cytometry of vesicle contents for the same line of cells. Comparative quantification of transmitters released during individual exocytosis events, using amperometry, and the total contents of individual vesicles, using cytometry at PC12 cells suggested that approximately 40% of the total dopamine content was released during a single exocytosis event. Partial exocytosis is not only possible, but it is highly likely that it is the primary mode of exocytosis with only a small number of amperometric events releasing large quantities of dopamine in full ejection of the vesicle content.
What Happens After Exocytosis
In most cases, the secretory vesicle cannot be recovered in its full form to be reused, as it has bonded with the cell membrane. The creation of vesicles is an energy-intensive process, and requires significant effort by the mitochondria, the energy factories in cells, to carry out this process. In non-constitutive exocytosis, such as neuronal exocytosis, where neurotransmitters are transported, the vesicles are not reused.
That being said, there are some forms of vesicle fusion that are not permanent, such as kiss and run fusion, which allows a partial deposit of vesicle contents, and then a reformation of the vesicle membrane. Recent research has also identified partial emptying of vesicles in some cases, where a secretory vesicle will release some of its contents, detach and reseal, and float back into the intracellular space. It can then return to the membrane and release the rest of its contents, or be re-filled with a new molecule at a receptor site for another organelle.
You May Like: Automatic Processes Definition Psychology
Calcium Sensors For Endocytosis
Recent studies show that calmodulin, a calcium-binding protein, is involved in rapid, slow, and bulk endocytosis and in endocytosis overshoot, whereas synaptotagmin, another calcium-binding protein, is involved in slow endocytosis. Thus, calmodulin and synaptotagmin may act as calcium sensors for endocytosis.
Calmodulin
Various calmodulin inhibitors block rapid, slow, and bulk endocytosis and endocytosis overshoot at calyces . They also block rapid endocytosis in chromaffin cells . Knockdown of calmodulin inhibits slow endocytosis at cultured hippocampal synapses . These results suggest that calmodulin is the calcium sensor for all forms of endocytosis .
Calcineurin
One of the downstream targets of calmodulin is calcineurin, which may dephosphorylate many endocytic proteins . Calcineurin is composed of a catalytic A subunit and a regulatory B subunit. Among the three isoforms of the A subunit, calcineurin A and A are expressed in the brain. Knockout of calcineurin A inhibits rapid and slow endocytosis at calyces , whereas knockout of calcineurin A inhibits slow endocytosis at hippocampal synapses . These results suggest the involvement of calcineurin in both rapid and slow endocytosis . The downstream target for calcineurin may include dynamin 1 because dynamin 1 dephosphorylation by calcineurin may be involved in bulk endocytosis .
Synaptotagmin
Comparison between calmodulin and synaptotagmin
Traditional Models Of Exocytosis
Until the last decade, exocytosis has generally been thought to be carried out via a full distention of the vesicle membrane into the plasma membrane. The early electron microscopy experiments, patch clamp and fluorescence all led to models where the bulk release process was all or none. Ultrastructural studies for example show that omega structures exist, but the existence of these structures does not necessarily prove that full fusion is the major pathway. Later techniques followed the exocytotic process dynamically, allowing exocytosis to be monitored in terms of single events. In general these techniques either monitor the content that is released or the fate of the vesicle membrane during and following the event. In this section we extract the evidence for full distention from several of these studies, which use chromaffin cells as a model for exocytotic release.
Recommended Reading: What Does Abiotic Mean In Science
Exocytosis And Cell Mechanics
In order to avoid multiple full-collapse exocytosis events disrupting the size or shape of the cell, endocytosis rapidly removes and recycles the vesicle components from the membrane. However, this recycling and delivery of membrane by exocytosis can be used by the cell to enlarge the membrane and surface area during cell shape changes. This is particularly relevant to cells undergoing mechanical stress, which occurs during processes such as cytokinesis, phagocytosis and in wound repair.
The interaction between membrane tension, unfolding and trafficking has been demonstrated by a number of studies. In fibroblasts, the cell spreads and increases surface area initially by lamellipodia extension and unfolding of the membrane reservoir stored in folded microvilli and blebs. After these membrane stores are depleted, an increase in membrane tension activates the second stage of spreading, where a fresh supply of membrane is brought to the leading edge by exocytosis. These exocytic vesicles predominantly contain glycosylphosphatidylinositol-anchored proteins, and are trafficked to the membrane in a microtubule independent manner. During this slower process of spreading, exocytosis increases the plasma membrane area by around 50%. , , , . This sequential process of membrane unfolding followed by exocytosis also occurs to facilitate macrophage cell shape changes during phagocytosis .
What is Fast Endophilin-Mediated Endocytosis?
Micro/nanodomain Calcium Coupling Of Exo
Three sets of evidence suggest that more than a few micromolars of calcium at the calcium micro/nanodomain are needed to trigger slow endocytosis. First, endocytosis at 0.50.75 M calcium is extremely slow, with a of ~600 s . Second, EGTA slows down endocytosis , whereas BAPTA, a faster calcium buffer, abolishes endocytosis . This result suggests that, analogous to the sensitivity of exocytosis to BAPTA and EGTA , calcium influx triggers not only exocytosis but also endocytosis at the microdomain near calcium channels , whose calcium concentration is more than 10 M . As the calyx matures, the coupling between calcium and exocytosis and that between calcium and endocytosis change from the microdomain to the nanodomain . Third, photolysis of a caged calcium induces endocytosis when the calcium concentration is more than ~10 M . These results suggest that the endocytosis trigger takes place at or near the release site, challenging a seemingly established principle that endocytosis takes place at the periactive zone . Consistent with this suggestion, FM dye uptake at snake neuromuscular junctions is near the active zone , a retrievable vesicle pool at the plasma membrane of hippocampal boutons was implied to be within ~100300 nm of the active zone , clathrin may be located at Drosophila active zones , and endocytic sites are within 500 nm of exocytic sites in PC12 cells .
You May Like: Cool Math Games Date Launched
Pore Opening And Full Fusion
The large fusion pore observed, on the other hand, may represent an intermediate before full collapse of vesicles. A secretory vesicle contains 6070 copies of Syb2, of which 13 are used during the fusion process. All of these Syb2 molecules, as well as other vesicular proteins such as synaptotagmin, are completely lost on the plasma membrane during the full collapse of the vesicle. Clathrin-mediated endocytosis must be initiated to collect and precisely recycle these vesicular membrane components rapidly. Originally believed to be a slow process, we find that the migration of preformed clathrin-mediate pits on the plasma membrane to the vesicle release sites is key to the clearance of exocytic slots in a timely fashion , which may explain the fast clathrin-mediated endocytosis observed in neurons .
Sequential Fusion And Multivesicular Exocytosis
After a KS fusion event, the invaginated fusion site before endocytosis can be targeted to harbor the next rounds of exocytosis , creating deep invaginations on the plasma membrane that may resulted in internalization of one large endocytic vesicle . These invaginations were initially found in non-excitable cells such as pancreatic acinar cells, mast cells, eosinophils and neutrophils and were later discovered in excitable cells such as pancreatic -cells and neurons . During sequential fusion, cis-SNARE complexes need to be removed from the fusion sites, and new trans-SNAREs on the plasma membrane must diffuse into these invagination structures. By adopting this configuration, vesicles that exist deep within the cytosol readily fuse with the plasma membrane to release their contents. This configuration also creates spatially preferred sites on the plasma membrane, conferring a mechanism for generating exocytosis hot spots in non-neuronal cells.
Don’t Miss: What Causes Parallax Error And How Do You Avoid It
Full Distention Of The Vesicle May Not Be Necessary For Quantal Release
In 2010, Amatore et al. studied the maximum aperture angle necessary for a vesicle to completely release its contents based on a physiochemical simulation of exocytosis and compared with several individual event spikes recorded by amperometry . The model was based on a vesicle fusing to the cell membrane and the consequences of constraining the membrane during release of vesicular contents, based on the radius and the angle of aperture for the vesicle. They determined that the vesicle only needs to open to about 10° in order to release its contents and provide the transient signal observed, as opposed to completely fusing with the cell membrane and fully dilating . This indicates that at the end of a release event the vesicle matrix is still mostly covered by membrane, and not exposed to the extracellular environment, suggesting that full fusion is unlikely for these events.
Fig. 10.Schematic membrane showing how the structural relationship of the vesicle with the membrane cytoskeleton network may constrain the maximum aperture angle .
Contribution Of Each Endocytosis Mode To Whole
Endocytosis is often evaluated as the averaged behavior of the entire cell or many boutons. The amplitude and time course of this behavior may depend on modes of endocytosis. We first introduce various averaged endocytosis behaviors, termed whole-cell endocytosis, and then discuss how different modes of endocytosis contribute to these behaviors.
Whole-cell endocytosis: rapid, slow, and overshoot
The kinetics of whole-cell endocytosis is highly plastic and depends on the stimulation intensity . The calyx of Held shows a typical example. At the calyx , a mild stimulus, such as an action potentiallike stimulus, induces a low level of exocytosis and rapid endocytosis with a time constant of ~13 s . An intermediate stimulus intensity, such as a short APlike train or a 20-ms depolarization, induces endocytosis with a of ~1030 s and bulk endocytosis . An intense stimulus, such as a prolonged, high-frequency APlike train or ten pulses of 20-ms depolarization at 10 Hz, reactivates rapid endocytosis together with slow endocytosis and bulk endocytosis . Finally, a very intense stimulation, such as a prolonged APlike train at very high frequency or ten pulses of 50-ms depolarization at 10 Hz, induces endocytosis overshoot that retrieves more vesicles than the exocytosed amount, together with rapid, slow, and bulk endocytosis .
Different whole-cell behaviors due to different endocytosis modes
Read Also: Open Sentence Definition Math