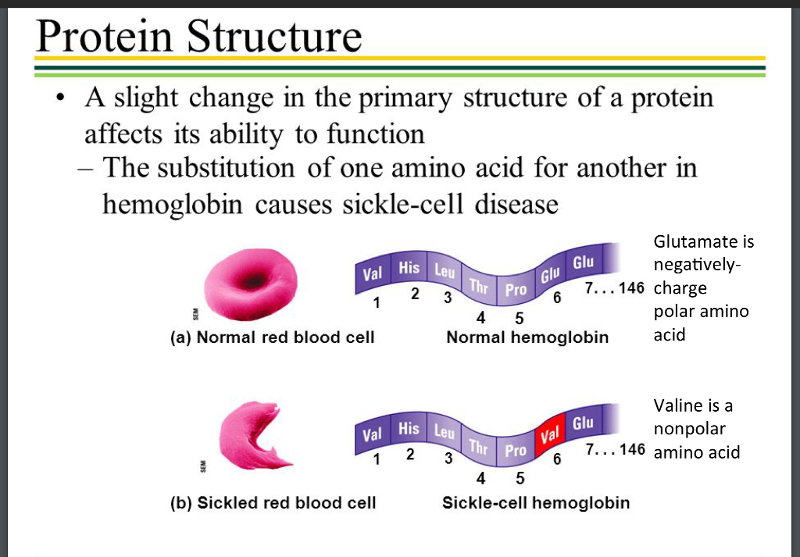
Protein Structure And Variety
Proteins are composed of chains of amino acids. A typical protein is about 400 amino acids long. As there are 20 different types of naturally occurring amino acids, many different proteins can be made. Different proteins are made of different combinations of amino acids.
The sequence of amino acids in the chain determines how the chain will fold up to make the protein, so different proteins have different three-dimensional shapes.
The three-dimensional shape of a protein determines its function. This is because proteins form attachments and interact with many other molecules and structures inside organisms.
The shape of a protein determines what it can interact with, just like the shape of a key determines which locks it can operate.
The Dna Sequences Of Entire Genomes Of Some Simple Organisms Will
The explosion in sequence data has just begun. DNA sequencing is far easier than protein sequencing, and the tools already available for cloning and efficient sequencing of 500-base-pair blocks of DNA will ensure that the current stream of new sequence data will become a torrent.
The ultimate target would be to determine the sequence of all the DNA in an organism, that is, to sequence an entire genome. Genomes range in size from 750,000 base pairs to more than 3 billion base pairs.
Such large-scale sequencing programs are feasible by today’s technology, but they are expensive in both manpower and actual dollar cost. Automated DNA sequencing techniques have begun to be developed, which should markedly diminish manpower requirements and decrease costs. It now seems likely that in the next few decades we will determine the complete DNA sequence of the bacterium Escherichia coil, the yeast Saccharomyces cerevisiae, the human genome, the fruitfly Drosophila, the mouse genome, the nematode Caenorhabditis elegans, and possibly even a number of plant and other bacterial and yeast genomes. The resulting information will stimulate future generations of biologists as they explore the functions of the tens of thousands of genes that will be revealed for the first time by such sequencing programs.
The Structure Of Macromolecules Is Governed By Foundational Principles Of Chemistry And Physics
The structure of macromolecules is governed by the foundational principles of chemistry and physics .
Associated learning goals
- Students should be able to relate basic principles of rate laws and equilibria to reactions and interactions and calculate appropriate thermodynamic parameters for reactions and interactions. A
- Students should be able to explain how a ligand, when introduced to a solution containing a macromolecule to which it can bind, interacts with the macromolecule. A
- Students should be able to explain, using basic principles, the effects of temperature on an enzyme catalyzed reaction. B
- Students should be able to discuss the dynamic properties of a macromolecule using foundational principles of physics. B
You May Like: What Happened To Beth Thomas Biological Father
Experimental Studies Search For Folding Intermediates
On the experimental side, much work has centered on the search for folding intermediates. Do the secondary structural elements in native proteins exist, as such, in small purified peptides apart from the rest of the structure? In the past it was thought that such structures would be so unstable that they would not be found, but recent evidence, based largely on optical spectroscopy, suggests a positive answer to the question, at least for certain sequences. Is there a definable folding pathway along which such structure intermediates can be found ? This question is controversial. Thermodynamic measurements can frequently be fitted to a two-state model reflecting only the native and the unfolded forms. Kinetic data, on the other hand, are often difficult or impossible to interpret without the assumption of one or more intermediate states. The methods are invariably indirect and the interpretation non-unique. Since crystals cannot be obtained for the unfolded state, x-ray diffraction data are not even potentially available. Detailed NMR studies on long peptides are still on the horizon, but may in the future provide direct structural information on these intermediate states.
Schematic diagram of the folding process for an all helical protein. The reaction starts with an extended chain containing no permanent intrachain interactions. This proceeds to a hypothetical intermediate with fluctuating helical segments that occasionally
Computational Methods Allow Amino Acid Sequences To Be Threaded Into Known Protein Folds
We know that there are an enormous number of ways to make proteins with the same three-dimensional structure, and thatover evolutionary timerandom mutations can cause sequences to change without a major change in the of a . For this reason, one current goal of structural biologists is to determine all the different protein folds that proteins have in nature, and to devise computer-based methods to test the amino acid sequence of a to identify which one of these previously conformations the domain is likely to adopt.
A computational technique called threading can be used to fit an sequence to a particular fold. For each possible fold known, the computer searches for the best fit of the particular amino acid sequence to that structure. Are the hydrophobic residues on the inside? Are the sequences with a strong propensity to form an in an helix? And so on. The best fit gets a numerical score reflecting the estimated stability of the structure.
Don’t Miss: Mcdougal Littell Geometry Teacher Edition Answers
Digestive And Excretory System
A fishs digestive and excretory system includes the structures and organs that bring food into the body, break food down into usable substances organism, and remove unused food. The digestive system begins with the mouth and teeth, which trap food and help send it on to the stomach and intestine for digestion. Undigested food and waste leaves the body through the anus .
The urinary portion of the excretory system also removes waste produced by the body. Its chief organs are the kidneys, which are a pair of long, dark-red organs under the vertebrae. The kidneys filter small molecules from the blood. After filtering, usable materials such as sugars, salts, and water are absorbed back into the blood. The remaining waste products pass from the kidneys down the urinary tubes, to the bladder, and out through an opening behind the anus, called the urogenital opening. This is the same opening through which materials from the reproductive system pass.
The gills are also part of the excretory system. Blood carries waste products and excess salts to the gill filaments. Carbon dioxide and ammonia are excreted by the gills. Fish living in seawater and brackish water also excrete excess salt from their gills.
The liver also removes wastes from the blood. The liver cleans blood after it has picked up digested products from the intestine. Wastes are converted into bile and stored in the gall bladder, where they wait to be poured back into the digestive tract to aid in digestion .
How Does The Structure Of An Enzyme Affect Its Function
4.9/5enzymestheir functiontheir structureenzymeenzyme
Subsequently, one may also ask, how does the structure of an enzyme affect its function quizlet?
Each enzyme has an area called an active site, this is where a substrate bonds and reacts with the enzyme. The binding of the correct molecule/substrate causes the enzyme to become active and perform its function.
One may also ask, how does protein structure affect its function? Protein structure depends on its amino acid sequence and local, low-energy chemical bonds between atoms in both the polypeptide backbone and in amino acid side chains. Protein structure plays a key role in its function if a protein loses its shape at any structural level, it may no longer be functional.
Also Know, why is the shape of an enzyme important to its function?
Each different type of enzyme will usually catalyse one biological reaction. Enzymes are specific because different enzymes have different shaped active sites. The shape of an enzyme’s active site is complementary to the shape of its specific substrate or substrates. This means they can fit together.
How does the shape of an enzyme determine its function?
An enzyme has a unique 3D shape b/c it is a protein. What does the shape of an enzyme determine? The shape of the enzyme determines which chemical reaction it will speed up. The active site of an enzyme only fits one type of substrate molecule.
Recommended Reading: Is Chemistry Or Physics Harder
A Technical Breakthrough Promises Information About Dynamic Processes In The Function Of Proteins
The massive electron-storage rings that physicists use to probe the fundamental components of matter also emit x-ray beams high in power. These synchrotron x-ray sources have recently been used to study large biological molecules. The beams of x-rays are thousands of times as strong as those from conventional laboratory x-ray sources, reducing x-ray data-collection time from months to hours. An experimental breakthrough in the application of multiple-wavelength x-ray diffraction now provides exposure times of milliseconds. The biochemical events on the surface of a protein can therefore be studied by a series of snapshots of the structure every few milliseconds. This should allow the sequence of events that constitute a chemical reaction or protein conformational change to be understood in atomic detail. Examining the dynamics of fundamental biological reactions will deepen our understanding of how proteins work, provide insight into normal functions, and raise the possibility of understanding abnormal functioning in disease.
Cell Structure And Function Overview
This section dives further into organelles and divides them into two functional groups organelles that maintain and repair the cell, and organelles that function to collect and distribute the energy needed for biochemical reactions. Lets start with organelles that maintain and repair cellular components.
Whether it is a prokaryotic organism or a eukaryotic organism, there is an ever-changing laundry list of tasks that must be completed in order for an organism to survive, grow, and reproduce. Before cells can replicate their DNA, they first need to collect and digest an energy source. This means the cell may need to move around, grow in size, fight off predators, or change environmental conditions. In a multicellular organism, even more functions are needed to prepare the organism for reproduction.
If we look at the cell membrane alone, we can start to understand the types of repair and maintenance tasks that need to be conducted. For instance, the phospholipids that create a cell membrane are constantly breaking down. These need to be replaced, and many more need to be created as a cell grows. Further, the many proteins embedded within the membrane and the other molecules that cells use to recognize each other, attach to surfaces, and communicate, also need to be created, repaired, or replaced almost constantly!
Together, these two organelles provide energy for almost all of the life on Earth in one way or another!
About
Don’t Miss: Geometry Basics Segment Addition Postulate Worksheet Answer Key
Structure And Function Of Biomolecules
| Paper Type: Free Essay |
All Biomolecules have certain functions and these molecules all have a unique structure which is why they function in these ways. These molecules are known collectively as macromolecules, these molecules are grouped into four main categories which each have their own structure. These structures are the key to the macromolecules functions as each of them do a specific task in the body.
If you need assistance with writing your essay, our professional essay writing service is here to help!
Macromolecules are grouped into carbohydrates, nucleic acids, proteins, and lipids. In most cases macromolecules are polymers, which is a long molecule which are made by linking together a large number of small, similar compounds called monomers. Polymers are formed by a dehydration reaction, this happens by the -OH group being removed from one monomer, and a hydrogen atom is removed from the other monomer, then the polymer is formed .
The Helix And The Sheet Are Common Folding Patterns
When the three-dimensional structures of many different molecules are compared, it becomes clear that, although the overall of each protein is unique, two regular folding patterns are often found in parts of them. Both patterns were discovered about 50 years ago from studies of hair and silk. The first folding pattern to be discovered, called the , was found in the protein –, which is abundant in skin and its derivativessuch as hair, nails, and horns. Within a year of the discovery of the helix, a second folded structure, called a , was found in the protein fibroin, the major constituent of silk. These two patterns are particularly common because they result from hydrogen-bonding between the NH and C=O groups in the , without involving the side chains of the amino acids. Thus, they can be formed by many different sequences. In each case, the protein chain adopts a regular, repeating conformation. These two conformations, as well as the abbreviations that are used to denote them in ribbon models of proteins, are shown in .
The regular conformation of the polypeptide backbone observed in the helix and the sheet. The helix. The NH of every peptide bond is hydrogen-bonded to the C=O of a neighboring peptide bond located
Two types of sheet structures. An antiparallel sheet . A parallel sheet. Both of these structures are common in proteins.
Recommended Reading: Parallax Errors
The Folding Problem Now Seems Ripe For Major Advances
The immediate future for the folding problem looks remarkably bright. The development of both fundamental and ad hoc theoretical approaches is advancing rapidly. The correlation and interactions between theory and experiment will be much closer than has generally been true in the past. Combined approaches, with various levels of theory or theory and experiment, seem likely to be the most fruitful. The ability to easily synthesize specific polymers, themselves specifically designed to test theoretical predictions or to provide missing values for parameters, seems particularly promising.
Instrumentation. The solution of the structures of new proteins, and of mutant versions of older proteins, will continue to be of major importance. Thus the development and implementation of new and improved x-ray and neutron diffraction procedures is as important to the folding problem it is as to other areas in structural biology. Improvements in both solid-state and high-resolution NMR will be central to the specification of the unfolded state and the search for definable folding intermediates. Proteins that are isotopically labeled at specific sites will be essential in this process, and they will also permit the study by NMR of substantially larger proteins than can currently be tackled.
The Structure/function Core Concept
Three published descriptions of the structure/function core concept can be found in .
Table 2. The core concept of structure/function
| Core Concept |
|---|
| From Vision and Change Report |
| 2. STRUCTURE AND FUNCTION: Basic units of structure define the function of all living things. |
| From Scientific Foundations for Future Physicians |
| Competency E5: Demonstrate knowledge of how biomolecules contribute to the structure and function of cells.Competency E6: Apply understanding of principles of how molecular and cell assemblies, organs, and organisms develop structure and carry out function. |
| From Michael and McFarland : note that Michael and McFarland have used the linking symbol to signal the fact that the interaction between the structure and function runs in both directions. |
| The functions of molecules, cells, tissues, or organs are determined by their form . Structure and function are intrinsically related to each other. |
The similarities between these three statements are obvious, but each of them suffers from the same lack of any explicit reference to how this concept is to be applied in thinking about physiological mechanisms. As such, the term structure/function is simply a truism we must always understand the structure generating a function in order to fully understand that function. For example, to fully understand the pumping action of the heart you must understand the anatomy of the heart .
You May Like: Equation To Find Half Life
Macromolecular Structure Determines Function And Regulation
Students should be able to explain and apply core concepts of macromolecular structure and function, including the nature of biological macromolecules, their interaction with water, the relationship between structure and function, and frequently encountered mechanisms for regulating their function.The learning goals below are categorized as introductory A, intermediate B and upper C.
Nucleic Acid And Protein Sequence Data Are Accumulating Rapidly
The amount of available information on the primary structure of biological polymers is increasing at an astounding rate. Two decades ago we knew the nucleotide sequence of only a single small nucleic acid, the yeast alanine transfer RNA. We knew the amino acid sequence of fewer than 100 different types of proteins.
Today more than 18 million base pairs of DNA have been sequenced, and the data are accumulating at more than several million bases a year. The first completed sequences were research landmarks. Now sequences are appearing so rapidly that many research journals refuse to publish such information unless it has some particular novel or utilitarian aspects. Indeed, sequence data are currently accumulating faster than we can analyze them, and even faster than we can enter them into the data bases by existing methods.
You May Like: John Thomas Brother Of Beth Thomas
Rna Structure Is An More Challenging Area Of Research
The structure of RNA has been even more difficult to deal with than that of proteins and DNA. Only the smallest class, transfer RNA, has yielded any solved crystal structures. All the transfer RNAs turn out to be similar L-shaped molecules. This similarity is reflected in the cloverleaf model for secondary structure, originally derived by searching for similar base pairing possibilities within the single chains. Although no other RNA structures are yet available through diffraction procedures, the extensive use of sequence data and sequence homology has led to a large array of secondary structure predictions that will almost certainly be retained in the three-dimensional structures eventually determined. Nuclear magnetic resonance is starting to provide a substantial amount of structural information on RNAs, but diffraction-quality crystals would be enormously useful.